Stem cells can generate organ-like structures, and in a handful of cases they contribute to functional tissue that works in a living body. But can they grow a fully formed, fully wired, fully vascularized whole human organ from scratch? Not yet, not reliably, and not outside highly specialized research labs. That's the honest answer. What they can do is already remarkable, and understanding the gap between what's possible now and what's still out of reach is the most useful thing you can take away from this article.
Can Stem Cells Grow Organs? What’s Possible and How
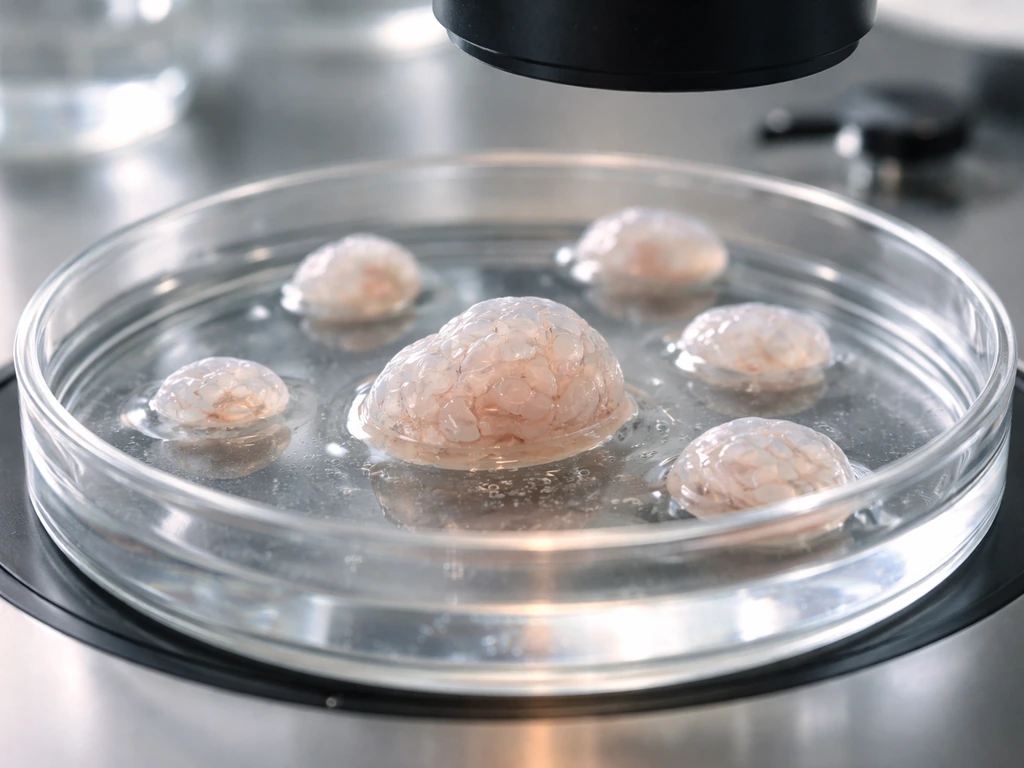
What 'growing an organ' actually means
This is worth pinning down because the phrase covers a huge range of things, from a few hundred cells in a dish to a transplantable kidney. There are three distinct levels researchers and science writers are usually talking about.
- Organoids: self-organizing, 3D clusters of cells (usually 0.1 to 1 mm across) that mimic the architecture and some functions of a real organ. They are not organs. They lack blood vessels, nerves, immune cells, and the full cell diversity of a mature organ.
- Engineered tissues: larger, sometimes scaffold-supported constructs grown in the lab. Examples include tracheal scaffolds seeded with a patient's own cells, or flat sheets of skin. These can be implanted but are typically simpler than whole organs.
- True whole organs: fully vascularized, innervated, immune-integrated structures at human scale with all the cellular complexity needed for long-term function. This is what most people mean when they say 'grow an organ,' and it is not yet broadly achievable.
If you want to understand whether we can truly grow organs in the fullest sense, the answer depends almost entirely on which of these three levels you mean. Organoids: yes, reliably. Engineered tissues: yes, for simple structures. Full whole organs: no, not yet.
Can stem cells make organ-like structures? Where research stands right now

Yes, and the progress over the last decade is genuinely impressive. Researchers have used both embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) to generate organoids representing the brain, liver, kidney, intestine, lung, heart, and more. iPSCs are particularly exciting because they are made by reprogramming a patient's own adult cells, which sidesteps immune rejection issues.
The NIH is taking this seriously enough that it is launching a dedicated Standardized Organoid Modeling (SOM) Center, specifically to establish validated, fit-for-purpose organoid protocols that replicate key structural and functional characteristics of real organs. The center's initial focus areas are liver, lung, heart, and intestine organoids. That kind of institutional investment signals that organoids are no longer a curiosity; they are becoming a standardized research tool. <a data-article-id="08DED660-D965-453B-AF47-3FBF75BF478BF">What scientists can currently grow in the lab is expanding rapidly, even if the full-organ finish line remains far off.
The challenge is that organoids, as useful as they are, are still developmental snapshots. They stall. They lack the blood supply needed to keep interior cells alive past a certain size. They don't mature the way a fetal organ does inside a body. Thinking about how cells in our organs actually grow during normal development helps clarify why replicating that process outside the body is so hard: it involves tightly choreographed signals, physical forces, and surrounding tissue interactions that are extremely difficult to reproduce in a dish.
How researchers guide stem cells toward organ fates
Getting a stem cell to become a liver cell instead of a neuron isn't magic, but it does require knowing the exact sequence of molecular instructions that a developing embryo uses. Think of it like a combination lock: you have to hit the right signals in the right order, or you get the wrong cell type or nothing useful at all.
Step 1: Getting the right starting material
You start with pluripotent stem cells, either ESCs or iPSCs. iPSCs are now the dominant choice for therapeutic research because they can be made from a patient's own skin or blood cells using just four transcription factors (the Yamanaka factors). This gives you a genetically matched starting material.
Step 2: Directing differentiation with signaling cues
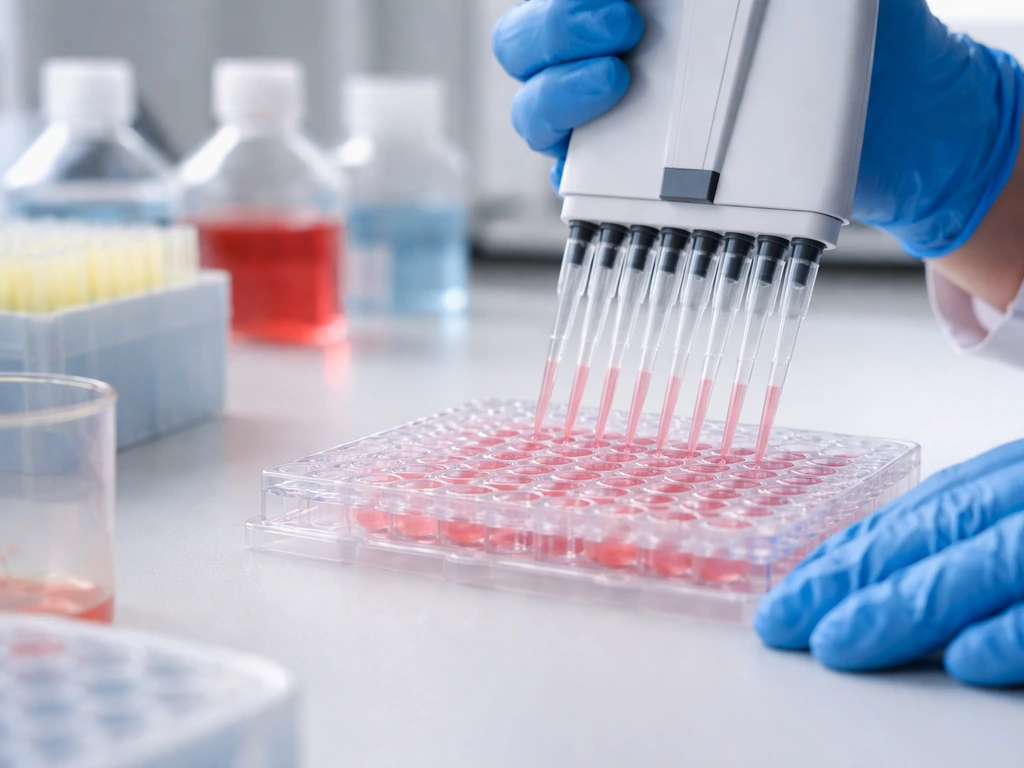
Researchers expose cells to carefully timed doses of growth factors, small molecules, and morphogens that mimic the signals an embryo produces. To make intestinal organoids, for example, you would apply Wnt signaling activators and inhibit BMP signaling at specific time points. Each organ type has its own protocol, and these protocols have been refined over years of trial and error.
Step 3: Creating a 3D environment
Cells grown flat on a plastic dish behave differently from cells in a 3D tissue. Most organoid protocols embed cells in a gel matrix (Matrigel is the most common) that provides mechanical support and lets cells self-organize into 3D structures. Bioengineered scaffolds made from collagen, fibrin, or synthetic polymers are used for larger engineered tissue constructs.
Step 4: Allowing self-organization
One of the most fascinating things about stem cells is that, given the right environment and signals, they will self-organize into patterns that resemble real tissue architecture. Intestinal organoids spontaneously form crypts and villi. Brain organoids develop layered cortical structures. This self-organization is driven by the same genetic programs that govern normal development. The cells, in a sense, remember what they are supposed to build.
Why building a true whole organ is still so hard
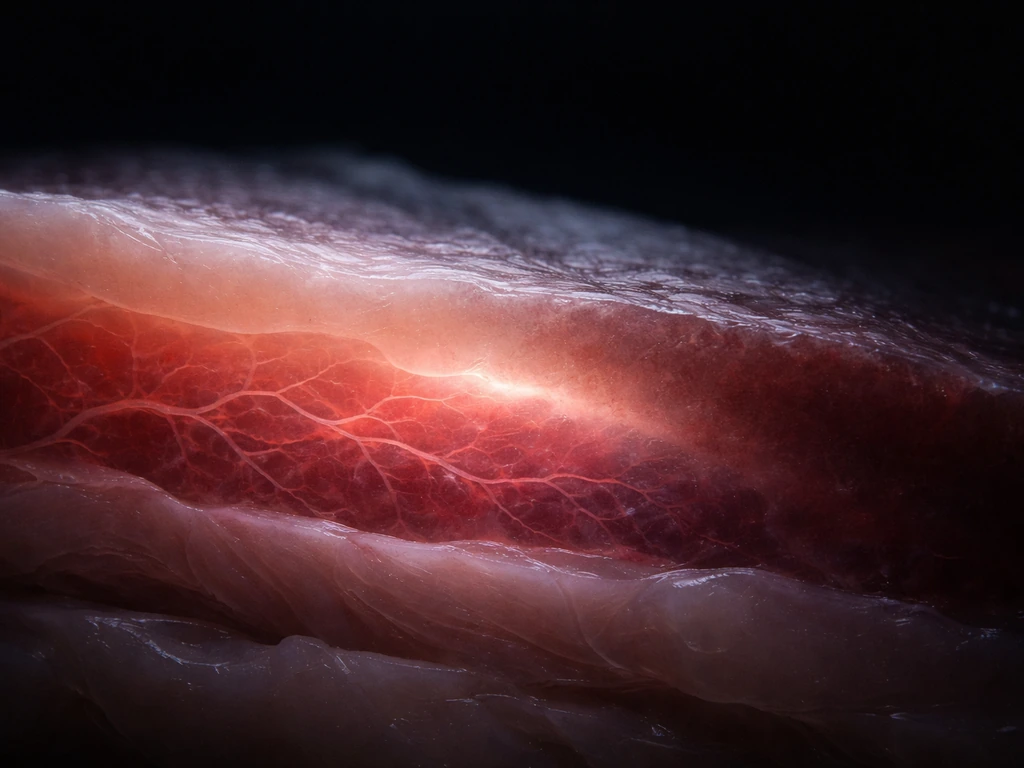
Even with all of the above done well, a true transplantable whole organ remains out of reach for most organ types. Here is where the real constraints live.
- Vascularization: any tissue thicker than about 150 to 200 micrometers needs a blood supply to deliver oxygen to interior cells. Building a branching network of blood vessels at the right scale and connecting it to a patient's circulation is one of the biggest unsolved problems in the field.
- Innervation: many organs need nerves to function properly. The gut, for instance, has its own enteric nervous system. Reproducing that wiring in a lab-grown organ is enormously complex.
- Scale: organoids are tiny. Scaling from a 0.5 mm organoid to a full-sized kidney (roughly 12 cm long) while maintaining viability, architecture, and function is not just a bigger version of the same problem. It is a categorically different problem.
- Maturation: lab-grown cells often stay in a fetal-like state and never fully mature into the adult cell types needed for organ function. A liver organoid may express fetal liver markers rather than the full metabolic machinery of an adult hepatocyte.
- Immune integration: a transplanted organ must be accepted by the immune system and integrated with surrounding tissue. Even with iPSCs from the patient's own cells, the epigenetic reprogramming process can introduce antigens that trigger immune responses.
- Reproducibility: organoid formation still involves significant batch-to-batch variability. Getting consistent results at scale is a major challenge, which is part of why the NIH's SOM Center initiative is so significant.
- Biomechanical environment: organs experience physical forces, blood pressure, fluid flow, stretch, and compression. Reproducing these forces in vitro and making tissue that responds to them correctly is an active area of research in bioreactor design.
What you can actually make today, and what it's useful for
Let's be concrete. Here is a realistic picture of where stem-cell-derived structures stand today across different organ systems.
| Organ Target | What's Achievable Now | Key Limitations | Clinical Use? |
|---|---|---|---|
| Intestine | Organoids with crypts/villi; used for drug testing and disease modeling | No vasculature, fetal maturation, no microbiome integration | Research only; some patient-derived models in clinical trials |
| Liver | Hepatic organoids; liver-on-a-chip systems; partial bioartificial liver devices | No full vascularization, incomplete adult metabolism | Bioartificial liver support devices in trials |
| Heart | Cardiac organoids; beating cardiomyocyte sheets; cardiac patches | No full-size vascularization, immature cardiomyocytes | Cardiac patches in early clinical trials |
| Kidney | Kidney organoids with nephron-like structures | No collecting duct connection, no filtration at scale | Research and disease modeling only |
| Lung | Airway and alveolar organoids; lung-on-a-chip | No full vascular tree, air-liquid interface challenges at scale | Drug testing and COVID-19 research |
| Brain | Cerebral organoids; region-specific organoids (cortex, hippocampus) | No vasculature, limited maturation, ethical concerns | Disease modeling and drug screening |
| Skin | Stratified skin grafts from iPSCs | No hair follicles, sweat glands in full constructs | Burn treatment; some grafts in clinical use |
| Trachea/Cartilage | Scaffold-seeded with patient cells | Limited to simple, avascular structures | Clinical implants performed (limited cases) |
The brain organoid space is particularly active and particularly nuanced. If you want to go deeper on what is and isn't feasible there, the question of whether you can grow a brain in a lab is worth exploring on its own, because the biological and ethical complexity is in a different category from other organ targets.
For a clearer sense of how scientists reliably produce enough cells to even run these experiments at scale, it helps to understand how researchers grow enough cells for their work, since cell expansion and quality control are fundamental bottlenecks that come before any organoid protocol even begins.
A practical roadmap: how you would attempt this today
If you are a researcher, student, or technically minded reader who wants to understand what attempting organ growth from stem cells actually looks like in practice, here is the high-level sequence. Think of each stage as a milestone with its own success criteria.
- Source your stem cells: Obtain iPSCs (reprogrammed from donor or patient cells) or ESCs (from approved cell line repositories). For NIH-funded work, you must use lines approved under the NIH Guidelines for Human Stem Cell Research. Success here means a stable, pluripotent, karyotypically normal cell line with confirmed expression of pluripotency markers (OCT4, SOX2, NANOG).
- Expand your cells: Scale up to the quantity you need using feeder-free culture systems and defined media. Success means consistent doubling time, no differentiation, and clean quality control metrics.
- Directed differentiation: Apply the published protocol for your target organ. For intestinal organoids this might be a 30 to 45 day stepwise process through definitive endoderm, hindgut, and intestinal stages. Success means expression of lineage-appropriate markers confirmed by immunostaining and RNA sequencing.
- 3D formation: Embed differentiating cells in Matrigel or an appropriate matrix and allow self-organization. Success means formation of recognizable 3D architecture (crypt-villus units, tubular structures, etc.) confirmed by histology.
- Maturation and culture: Maintain organoids in long-term culture, passaging to expand them. Success means stable growth, maintenance of architecture, and functional readouts (enzyme activity, barrier function, contractility, depending on organ type).
- Vascularization attempts (advanced): For larger constructs, attempt co-culture with endothelial cells or use microfluidic bioreactor systems to create perfusable channels. This is where most whole-organ attempts currently stall. Success at this stage would mean sustained cell viability in constructs thicker than 200 micrometers.
- Functional validation: Test whether your construct does what the organ is supposed to do. Liver constructs should metabolize drugs. Cardiac patches should contract rhythmically. Intestinal organoids should form a barrier and respond to pathogens. This is your primary success criterion.
- Scaling and reproducibility: Replicate results across multiple batches and, for therapeutic applications, across multiple donor lines. This is the stage where most promising results hit their hardest wall.
Realistic expectations: at steps 1 through 5, a well-resourced lab with existing expertise can expect to generate recognizable organoids within 4 to 8 weeks for most organ targets. Steps 6 through 8 are active research frontiers where no lab has a fully solved protocol. If your goal is a transplantable whole organ, you are working at the edge of what the field knows.
It is also worth knowing that one experimental approach involves growing human cells inside animal embryos, a strategy sometimes called chimera research. The debate around growing animals in artificial wombs overlaps with this space, since both involve creating biological environments for human or part-human tissue development. The ISSCR defines a chimera in stem cell research as an organism carrying cell populations from two or more genetically distinct sources, including cases where human stem cells are transferred into non-human embryos. NIH guidelines currently prohibit introducing human pluripotent cells into nonhuman primate blastocysts and restrict breeding of animals where human cells may have contributed to the germline.
Safety, ethics, and the regulatory landscape
This is not a minor footnote. If you are working with stem cells or organoids, or if you want to understand what's actually permitted, the regulatory and ethical environment shapes everything from which cell lines you can use to which experiments require special review.
Who regulates what
In the United States, NIH-funded research using human embryonic stem cells is governed by the NIH Guidelines for Human Stem Cell Research. These guidelines specify which hESC lines are approved for use and what oversight is required. iPSC research has fewer restrictions in many contexts but still falls under IRB (Institutional Review Board) oversight and informed consent requirements when derived from human donors.
The ISSCR publishes international guidelines that most research institutions follow globally. For specialized research areas including chimera experiments, the ISSCR recommends specialized review by ethics committees with relevant expertise, not just standard IRB review.
Key ethical questions in organoid research
Whether it is ethical to grow human organs in a lab is a question the field is actively working through, not one that has been settled. The main issues include: the moral status of organoids (especially brain organoids that develop electrical activity), informed consent for donor-derived iPSCs, equitable access to any therapies that emerge, and the boundaries of chimera research.
Brain organoids get the most ethical attention because some have shown spontaneous neural activity. Whether this constitutes anything like sentience or experience is debated, but it has prompted many research institutions to create specific oversight committees for brain organoid work. Most researchers in the field support this kind of specialized review as a responsible boundary-setting measure.
Safety considerations in engineered tissues
For any tissue intended for clinical use, safety requirements are extensive. Tumorigenicity (the risk that residual undifferentiated stem cells could form teratomas after transplant) is the primary concern. Protocols must include steps to remove or confirm the absence of undifferentiated cells before implantation. Manufacturing must meet Good Manufacturing Practice (GMP) standards. Clinical trials for stem-cell-derived tissues fall under FDA oversight in the US, specifically under the Center for Biologics Evaluation and Research (CBER).
One question that comes up naturally once you start thinking about transplanting lab-grown tissues is what happens to them after they go into a body. Whether transplanted organs continue to grow once implanted is a real concern, especially for pediatric patients, and the answer involves interesting biology about how organ size is regulated by systemic signals rather than just local cell division.
The straight answer and where to go from here
Stem cells can reliably generate organoids and simple engineered tissues that capture some of the structure and function of real organs. They cannot yet reliably produce fully formed, fully vascularized, correctly wired whole organs ready for transplant. That gap is real, and it is defined by specific solvable (but not yet solved) problems: vascularization, innervation, scale, maturation, and reproducibility.
The field is moving fast. The NIH's investment in standardizing organoid protocols for liver, lung, heart, and intestine suggests we are entering a phase where organoid science becomes more reproducible and clinically applicable. Whole-organ engineering is a longer horizon, but it's being actively worked on, not just theorized about.
If you want to follow this area closely, the ISSCR guidelines and NIH stem cell information pages are the best primary sources. For the underlying biology of how cells organize and grow into complex structures, understanding the basic rules of cell division, size limits, and developmental signaling will give you the framework to evaluate new research as it comes out rather than just reacting to headlines.
FAQ
If stem cells make organoids, why can’t that automatically become a transplantable organ?
No, “organoid” and “whole organ” are not the same. Organoids usually represent a developmental snapshot, often lacking a fully functional blood supply and nervous wiring, and they typically do not reach the full size, maturity, and organization needed for transplantation. The closer a product gets to an implantable organ, the more issues like vascular perfusion, innervation, and long-term integration become limiting.
What specifically prevents stem-cell-derived tissues from growing large enough to survive on their own?
Vascularization is the main size-limiting step. Many organoid protocols can create 3D structure, but interior cells become deprived of oxygen and nutrients as size increases. Researchers often need an additional strategy, such as engineering blood-vessel networks or using supportive microenvironments, to get beyond the early “diffusion-limited” stage.
When people say “wired organs,” what does that mean in practice, especially for brain tissue?
Wiring is more than “cells that look like neurons.” Functional integration requires correct, long-range connectivity, timing, and activity patterns, plus appropriate support glial and vascular interactions. This is why brain-related goals get treated as a distinct challenge area, with both technical and oversight requirements.
Why do stem-cell organoid results vary so much between labs?
One reason is reproducibility. Protocols may work in one lab and still fail elsewhere due to differences in starting cell quality, exact growth-factor timing, matrix composition, and quality control checkpoints. Standardization efforts (like validated organoid protocols) are meant to reduce variation so results can be compared across studies.
Does using a patient’s iPSCs mean immune rejection is solved?
iPSCs can sometimes reduce immune rejection risk because they can be made from a patient’s own cells, but immune compatibility is not guaranteed in every case. Even autologous cells can trigger immune issues due to differentiation state, culture-induced changes, or residual undesired cell types, so safety testing remains essential for any clinical intent.
What is the biggest safety hurdle for transplanting stem-cell-derived tissues?
Tumor risk is a major reason transplant is hard. If undifferentiated or partially differentiated cells remain, they can form teratomas after implantation. Good practice requires reliable methods to eliminate or confirm the absence of those cells, plus extensive release testing before any clinical use.
What changes when a promising lab protocol has to become a real clinical-grade product?
Even if growth is impressive in a dish, engineering for clinical use usually requires manufacturing controls that match GMP standards. That means robust, documented, batch-to-batch consistent processes, along with predefined acceptance criteria for identity, purity, potency, and contaminants, not just “it grew in the lab.”
If lab-grown tissue is transplanted, will it keep growing normally inside the body?
After implantation, organ size and function depend heavily on systemic signals (for example, hormones, growth regulators, and feedback from surrounding tissues) as well as the local tissue’s ability to integrate and receive sustained perfusion. For pediatric patients, these feedback dynamics and growth trajectories are especially important to plan for.
Are chimera experiments the only way to get more mature or vascularized organ development?
Yes, but the rules are strict and vary by jurisdiction and experiment type. In the US, human pluripotent cell work has specific federal guidance, and chimera-related research has additional oversight and restrictions. Institutions typically require specialized ethics review beyond standard IRB processes for chimera experiments.
What common media exaggeration should I watch for when I hear claims that “organs are growing”?
Not always, and it’s easy to misread headlines. The field sometimes demonstrates organoid-like features in a controlled setting, while full claims about function or transplant readiness require multiple layers of evidence: correct cell identities, structural organization, reproducible behavior, safety testing, and functional integration in relevant models.
What Controls When and How Fast Cells Grow and Divide
Learn what controls cell division timing and speed: checkpoints, signals, resources, DNA damage, stress, and failure lea


