Yes, scientists can grow organs in the laboratory, but with a very important asterisk: what exists today are tissue patches, miniature organ models called organoids, and bioengineered constructs, not fully transplant-ready whole organs. A working kidney you could just slot into a patient's body? Not yet. A tiny, self-organized cluster of liver cells that secretes albumin, responds to drug exposure, and mimics liver architecture? Absolutely, and that is genuinely useful. The gap between those two things is the story of where organ-growing science stands in 2026.
Can Scientists Grow Organs in the Lab? What’s Possible Now
What "growing organs" actually means today
When most people ask "can scientists grow organs," they are picturing a transplant-ready replacement heart or kidney assembled in a lab. That framing bundles together two very different goals. The first is growing functional tissue models: small, lab-made constructs that replicate enough of an organ's biology to be useful for research, drug testing, or localized repair. The second is fabricating a complete, vascularized, innervated, transplant-scale organ. Right now, science is genuinely succeeding at the first goal and making slow, real progress toward the second.
The word "organoid" comes up constantly in this field. The NCI defines an organoid as a 3D, mini-organ-like structure made by growing stem cells or a patient's own cells in the lab, one that contains multiple cell types and mimics aspects of the structure and some functions of the corresponding human tissue. Think of it as a biological scale model rather than the real building. It captures the layout and some of the behavior without being full size or fully connected to the body's supply lines.
A separate but related concept is the tissue-engineered construct: a larger, scaffold-supported piece of tissue grown to potentially repair or replace a damaged section of an organ (a skin graft, a trachea patch, a small section of bladder wall). These constructs are closer to clinical use than whole-organ fabrication and represent the most realistic near-term wins in the field. Understanding how far we can actually go when we try to grow organs requires keeping both definitions clearly in mind.
Where the field actually is right now
The honest state of the art looks like this: organoids and tissue patches are well-established, bioprinted constructs are advancing rapidly, and whole-organ fabrication remains a long-term research goal with genuine but incremental progress. NIH framed its 2025 organoid development initiative around liver, lung, heart, and intestine models, with the explicit aim of making those models reproducible and standardized enough for regulators to accept them. Notice that framing: the near-term translational emphasis is on better models and localized tissue products, not on printing a complete transplant-ready heart.
Skin is the current leader in clinical translation. The California Institute for Regenerative Medicine has described a GMP-compatible manufacturing platform that converts validated iPSCs (induced pluripotent stem cells, which are adult cells reprogrammed back to a stem-like state) into epidermal cells, dermal cells, and melanocyte precursors, producing autologous skin composite grafts. That is a real, quality-controlled, patient-specific product pipeline. A few steps behind skin are tracheal patches, bladder augmentation constructs, and corneal tissue. The NIBIB has noted that while more complex tissues like heart, lung, and liver have been recreated in the lab, they remain a long way from being fully functional transplant-ready organs.
Organoids have made the most noise scientifically. Brain organoids, kidney organoids, gut organoids, liver organoids: researchers can now produce miniature versions of most major organs from iPSCs. A 2023 study reported efficient, reproducible generation of expandable liver organoids from iPSCs, with albumin secretion used as a quality marker for differentiation. That is meaningful biology. But an albumin-secreting organoid the size of a rice grain is not a liver you can transplant.
If you want to understand the cellular machinery driving all of this, it helps to start with the basics of how cells in our organs actually grow and organize themselves, because that same biology both powers and limits what engineers can do in the lab.
How scientists try to build an organ: the four-part toolkit
Cell sources: where the raw material comes from

Every lab-grown tissue starts with cells. The two main inputs are adult stem cells (ASCs) taken directly from tissue samples and pluripotent stem cells (PSCs), which include embryonic stem cells and iPSCs. iPSCs are particularly powerful because they can be derived from a patient's own skin or blood cells, reprogrammed to a stem state, and then directed to become almost any cell type. This matters enormously for immune compatibility: a kidney organoid grown from your own iPSCs carries your genetic identity and is less likely to trigger rejection. The question of exactly how stem cells contribute to growing organ-like structures is one of the most active areas in the field right now.
Scaffolds and extracellular matrix: the architecture
Cells do not float freely in real organs. They are embedded in a protein-rich extracellular matrix (ECM) that provides structural support, chemical signals, and spatial cues. In the lab, researchers replicate this using hydrogels (fibrin and Matrigel are the most common), decellularized ECM from donor tissue, or synthetic polymer scaffolds. A 2024 review on decellularized ECM highlighted that scaffold choice directly affects biocompatibility and whether organoids can receive enough nutrients to fully mature. Decellularized scaffolds, made by stripping cells from a donor organ and leaving only the protein scaffold behind, are particularly promising for whole-organ engineering because they preserve the original vascular channels.
Bioprinting: laying cells down precisely

3D bioprinting uses a printer head loaded with "bioink" (a mixture of cells and hydrogel) to deposit living material layer by layer, like an inkjet printer but with biology instead of dye. The appeal is precision: you can theoretically print multiple cell types in the exact spatial arrangement found in a real organ, including the branching patterns of blood vessels. A 2022-2023 PMC study reported printing perfusable networks of synthetic capillary-scale 3D vessels, enabling transport in multi-cubic-millimeter tissue constructs. Printing the vasculature alongside the tissue is one of the most promising routes to solving the size problem (more on that shortly).
3D culture and organoid self-assembly
The other major approach does not try to engineer organ structure from the outside. Instead, it lets cells self-organize. When stem cells are placed in the right 3D environment with the right chemical signals, they spontaneously sort themselves into tissue-like arrangements. This is how most organoids are made. The result is less geometrically perfect than a bioprinted construct but often more biologically realistic because the cells are following their natural developmental programming. Researchers are now also exploring how to scale up cell production reliably, a challenge well described in work on how scientists grow enough cells for their experiments in the first place.
The four walls you keep running into

Growing a clump of cells is relatively straightforward. Growing a functional organ-scale tissue is not. Four constraints keep coming up in the literature, and they are worth understanding individually because each one maps to a specific line of current research.
Vascularization: the oxygen problem
This is the biggest bottleneck in organ engineering, and it has a clean physical explanation. Oxygen diffuses through tissue roughly 100 to 200 micrometers before it is consumed. Beyond that distance, cells suffocate. A real organ gets around this by having a dense network of capillaries that deliver oxygen to every cell within that diffusion range. Lab-grown tissues mostly do not have that. Without a perfusable vascular network, any tissue thicker than about 200 micrometers develops a necrotic (dead cell) core. Multiple reviews and studies confirm this 100-200 µm diffusion constraint as the central physical barrier to scaling up tissue constructs.
Researchers are attacking this in two ways. First, bioprinting vascular channels directly into constructs, targeting vessel diameters in the 100 to 1,000 µm range to match transport needs for millimeter-scale tissues. Second, using microfluidic organ-on-chip platforms that route culture media through printed channels to simulate blood flow. A 2024 Nature Communications study described a perfusable on-chip vascular network that supported hiPSC-derived lung organoid growth over roughly two weeks, without requiring transplantation into a living animal. For kidney organoids specifically, a 2024 Biofabrication paper from a Harvard-affiliated group reported a perfusable, vascularized kidney organoid-on-chip model using microvessel design and perfusion readouts. These are real advances, but they are working at the millimeter scale, not at the fist-sized scale of a human kidney.
Maturation: getting cells to grow up
Even when you solve the immediate oxygen problem, lab-grown cells tend to stay in an immature, fetal-like state. A 2022 study on kidney organoids made this explicit: iPSC-derived kidney organoids lack functional vasculature in vitro, and the sparse endothelial cells present tend to be lost during prolonged culture. This restricts the organoid's ability to mature into adult-type kidney tissue. The same pattern appears across organ types. Maturation requires the right mechanical forces, hormonal signals, and cell-cell interactions that are hard to fully replicate outside a living body. Some researchers transplant organoids into animal hosts to let the host's circulation and signaling environment drive maturation, which works but adds complexity and raises questions about scalability.
Immune compatibility: will the body accept it
Even a perfectly engineered organ could be rejected if the immune system treats it as foreign tissue. Using the patient's own iPSCs to generate the organ largely solves this, but iPSC-derived products are not perfectly invisible to the immune system, partly because the reprogramming and differentiation process can introduce epigenetic changes and stress markers. For scaffold-based approaches using decellularized donor tissue, residual donor antigens in the matrix can still trigger immune responses. And any animal-derived scaffold materials, like Matrigel, introduce safety and regulatory complications that the FDA's April 2024 guidance on human- and animal-derived materials in manufacturing directly addresses.
Functional integration: does it actually connect
An organ is not just tissue, it is tissue that connects to nerves, blood vessels, lymphatics, and neighboring organs in a specific way. A lab-grown liver construct needs to hook up to the portal vein, hepatic artery, bile ducts, and hepatic veins to function. A transplanted kidney needs to connect to the ureter and renal vasculature. Getting the vascular inlets and outlets to anastomose (surgically join) with the patient's own vessels without leaking or clotting is a major surgical and bioengineering challenge. A 2024 Frontiers review noted that connection of engrafted kidney constructs to systemic circulation can be demonstrated within about two weeks after engraftment in some animal model experiments, which is encouraging, but animal model success is a long distance from human clinical use. This question also connects to a broader one: what actually happens to transplanted organs once they are placed in a new body and whether they continue to grow and integrate normally.
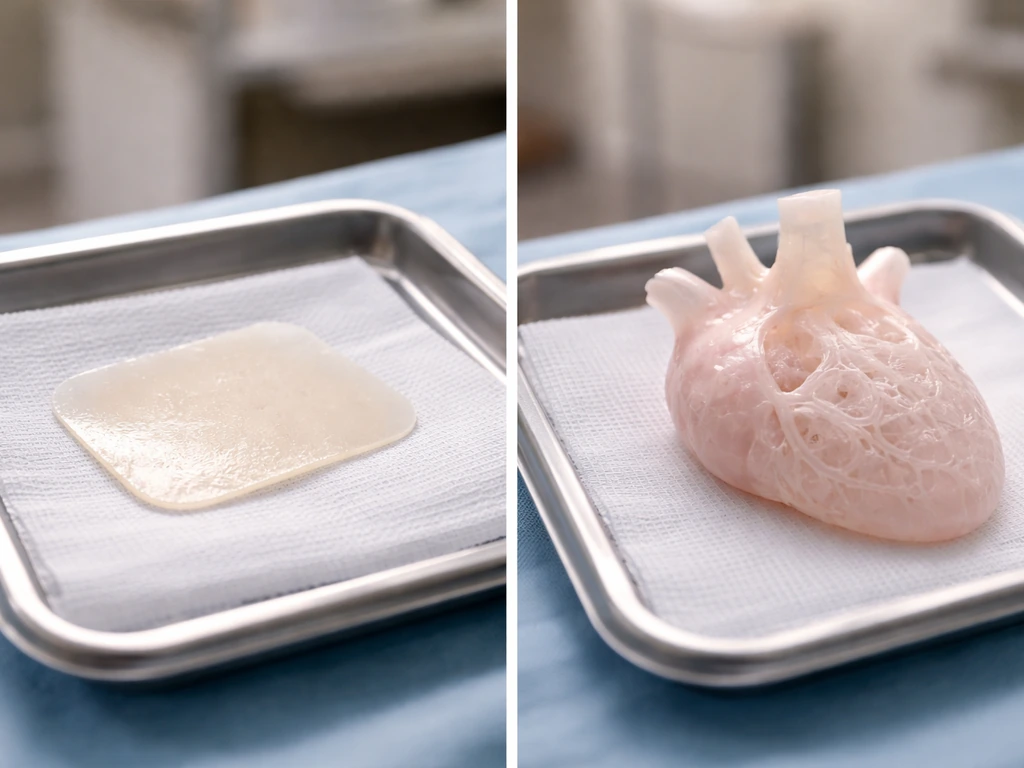
What is realistic now versus what will take decades
| Application | Current status | Realistic timeline |
|---|---|---|
| Skin grafts (iPSC-derived) | GMP-compatible platforms exist; clinical trials underway | Now to near-term |
| Trachea/bladder patches | Small constructs implanted in humans experimentally | Near-term with more data |
| Organoids for drug testing/modeling | Widely used; NIH standardization push ongoing (2025) | Now |
| Organ-on-a-chip platforms | Active research; perfusable vascular networks demonstrated | Now for research; years for diagnostics |
| Bioprinted tissue patches (heart, liver) | Millimeter-scale constructs with basic function demonstrated | 5-15 years for clinical use |
| Whole transplant-ready kidney or liver | No functional whole organ demonstrated at human scale | Decades away, timelines highly uncertain |
| Whole transplant-ready heart or lung | No functional whole organ demonstrated at human scale | Decades away, timelines highly uncertain |
The honest answer is that the closer an application is to a flat, thin, or simple tissue, the more achievable it is right now. Skin is thin and does not need internal vasculature to survive as a graft. A tracheal patch needs structural support but not a complex internal blood supply. Move to a thick, metabolically demanding organ like a liver or heart and every one of the four constraints above hits simultaneously. NIBIB's tissue engineering resources put it plainly: complex organ tissues remain a long way from being fully transplant-ready. That is not pessimism, it is an honest reading of the physics and biology.
One area that draws particular curiosity is neural tissue. Growing brain-like structures in a lab is genuinely possible at the organoid scale, and brain organoids are already being used to study neurological diseases. But a transplant-ready brain construct raises a separate set of biological and philosophical questions that go far beyond vascularization.
Safety, ethics, and the regulatory path to patients
Even when a lab-grown tissue works in animal models, getting it to patients requires navigating a detailed regulatory framework. In the United States, the FDA regulates tissue-engineered medical products and cell therapies under its regenerative medicine framework, which includes the Regenerative Medicine Advanced Therapy (RMAT) designation. RMAT is an accelerated pathway for products showing preliminary clinical evidence of addressing serious unmet medical needs, but "accelerated" still means rigorous premarket evidence, not a shortcut. The FDA's framework also makes clear that the specific pathway depends on the product: a bioengineered skin graft is regulated differently than a cell-infused organoid intended for internal transplantation.
Quality control is a real and underappreciated challenge. A 2022 quality discussion paper noted that organoid-relevant cell products require identity checks, genetic stability testing (karyotyping), and contamination screening including mycoplasma detection. Cells grown in culture accumulate mutations; an iPSC line that started healthy can develop genomic abnormalities after extensive expansion. The NIH's 2025 organoid center initiative was specifically designed to address reproducibility: organoids made in one lab often look and behave differently from those made in another, which makes regulatory acceptance harder.
The ethical side of this work is genuinely complex, and it is not limited to abstract philosophy. The ISSCR's 2021 updated guidelines address ethics and clinical translation considerations for stem cell-derived constructs, including cautions around genome editing and high-risk applications. They call for substantial preclinical evidence before any clinical application. One question that comes up often: if you grow an organ from a patient's own cells, who owns it? What oversight exists if that organ is modified genetically? These are live regulatory and ethical questions, not hypothetical ones. If you want to think through where the ethical lines are being drawn, the ethics of growing human organs is a topic worth exploring carefully. And adjacent to that is the broader question of how society governs related biotechnologies: debates about growing animals in artificial wombs share many of the same governance challenges around consent, personhood, and scientific responsibility.
What you can do right now if this field interests you
Whether you are a student, an educator, or someone thinking about a research career, there are concrete and practical ways to engage with this field today. The concepts you need to understand first are not exotic: cell division and mitosis, differentiation, extracellular matrix biology, and basic transport physiology (how oxygen and nutrients move through tissue). Those foundations explain why every constraint described above exists, and they are covered in standard cell biology and physiology courses.
- Learn the core biology first: mitosis, cell differentiation, stem cell types (ASC vs PSC vs iPSC), and ECM structure. These concepts directly explain both the capabilities and the limits of organ engineering.
- Follow primary sources: Nature Methods, Nature Biomedical Engineering, Biofabrication, and Biomaterials publish the most relevant primary research. NIH Research Matters and NIBIB's fact sheets are good plain-language summaries.
- Understand the regulatory landscape: read the FDA's regenerative medicine framework page and the RMAT designation description. Knowing how products get to patients is as important as the science for anyone considering a career in this field.
- Look for organoid or tissue engineering courses: many universities now offer lab modules in organoid culture, and online platforms such as Coursera and edX have biofabrication courses. Hands-on work with hydrogels and cell culture changes how you think about the scale and texture of the problems.
- Evaluate claims critically: when a headline says "scientists grow [organ]", ask whether they mean a whole transplant-ready organ or an organoid/tissue patch. The distinction matters enormously. Check whether the study was done in vitro, in an animal model, or in a human clinical trial.
- Consider adjacent entry points: tissue engineering, biomaterials science, stem cell biology, and biomedical engineering are all routes into this work. Computational modeling of oxygen transport and vascular network design is an increasingly valuable skill set that is accessible to people with a physics or math background.
If you are a student trying to design a class project or science fair experiment around this topic, the most tractable entry points are observing 3D cell culture behavior, modeling oxygen diffusion mathematically, or examining how scaffold material (agar vs gelatin vs fibrin) affects cell growth patterns. You do not need a full bioprinting setup to engage meaningfully with the underlying biology.
The bottom line: scientists can and do grow organs in a meaningful sense today. The tissues and organoids coming out of labs right now are already changing how drugs are tested, how diseases are modeled, and how some patients receive care. Full transplant-ready whole organs are a genuine long-term goal with real scientific momentum behind it, but the vascularization problem, the maturation problem, and the integration problem are physical and biological constraints, not just engineering puzzles waiting for a clever fix. Watching how researchers work around those constraints, step by step, is one of the more fascinating scientific stories of the next few decades.
FAQ
If scientists can grow organoids, why can’t they just transplant them like whole organs?
Usually, no. Most current “organ-growing” products are intended for research use or local tissue repair, and they do not yet meet the combined requirements for long-term survival in a human body (including reliable perfusion, mature function, and safe integration).
What actually limits how big an organoid can get?
Organoids often lack a durable, fully functional vasculature, so they can grow only to a limited size before oxygen and nutrient limits cause the inner region to fail. Some groups use organoid-on-chip systems to improve transport, but that is still not the same as connecting to a patient’s blood vessels.
Are hydrogels like Matrigel and decellularized ECM equally suitable for building organ-like tissues?
A common mix-up is treating “Matrigel” and decellularized scaffolds as interchangeable. Matrigel is animal-derived and can complicate manufacturing and regulatory approval, while decellularized ECM aims to preserve native structural cues and, in some designs, the original vascular channels.
If the cells come from a patient, will the immune system always accept the grown tissue?
Yes, but it can also reduce it. Using iPSCs made from the patient typically improves immune compatibility, yet immunogenicity can still arise from culture-induced stress, epigenetic shifts, or residual donor antigens in scaffold materials. This is why immune testing and risk assessment still matter even for autologous approaches.
What quality checks are typically required before lab-grown tissue can be considered for patients?
To call something “clinical-grade,” teams must run identity and safety tests on the cell source and final product. In practice that includes checks such as mycoplasma screening, genetic stability monitoring (since cultured cells can accumulate changes), and batch-to-batch reproducibility measures.
How do organoids used for drug testing differ from tissue constructs aimed at repairing injuries?
They can play different roles. iPSC-derived organoids are often used to model disease and test drugs, while tissue-engineered constructs are often targeted at repairing a specific damaged area. Whole-organ transplantation requires extra steps (scale, maturity, and reliable vascular and nerve integration) that are not yet part of routine workflows.
What measurements show that an organoid is functioning more like the real organ?
Your readout can matter as much as the organoid itself. For example, albumin secretion is used as a differentiation and functional signal in liver organoid work, but researchers usually combine marker panels with functional assays to avoid being misled by partial or abnormal differentiation.
Why do organoids from different labs sometimes look and behave differently?
Not always. Some organoid protocols yield consistent “disease-like” behavior, but other steps such as starting cell quality, differentiation timing, and scaffold composition can change outcomes. That is why reproducibility and standardized protocols are emphasized before regulatory review becomes realistic.
Can I do a safe, realistic organoid experiment as a class project without needing advanced equipment?
Yes, and the safest strategy depends on the intended application. For student or early research settings, simpler 3D culture or diffusion modeling projects can still be valuable, but attempting genome editing or high-risk patient-facing applications requires specialized oversight and evidence far beyond standard coursework.
Which kinds of tissues are the most likely to reach patients first, and why?
Right now, the most believable near-term pathway is localized repair with predictable structure, not replacing a whole organ. If the goal is clinical translation, “thick, highly metabolic, and tightly connected to blood and nerves” tissues are currently the hardest, so progress is usually staged by complexity.
After a transplant works in animals, what still has to be proven for human success?
The main red flags are stability over time and reliable integration. Even if function looks promising shortly after implantation in animal models, teams must check long-term performance, vascular patency, immune reactions, and whether the tissue continues to mature appropriately.
Can We Grow Organs? Science, Limits, and What Comes Next
Can we grow organs? See what’s possible now with organoids and tissue patches, plus limits like vessels, nerves, and sca