Yes, we can grow organs, but with a big asterisk. Right now, scientists can grow small, simplified organ-like structures called organoids, engineer tissue patches that work in patients, and even transplant lab-grown grafts into human eyes. What we cannot do yet is reliably grow a full-sized, fully functional, vascularized organ ready for transplant, like a kidney or liver, entirely outside the body. That gap between what's possible today and what the headlines sometimes promise is exactly what this article unpacks.
Can We Grow Organs? Science, Limits, and What Comes Next
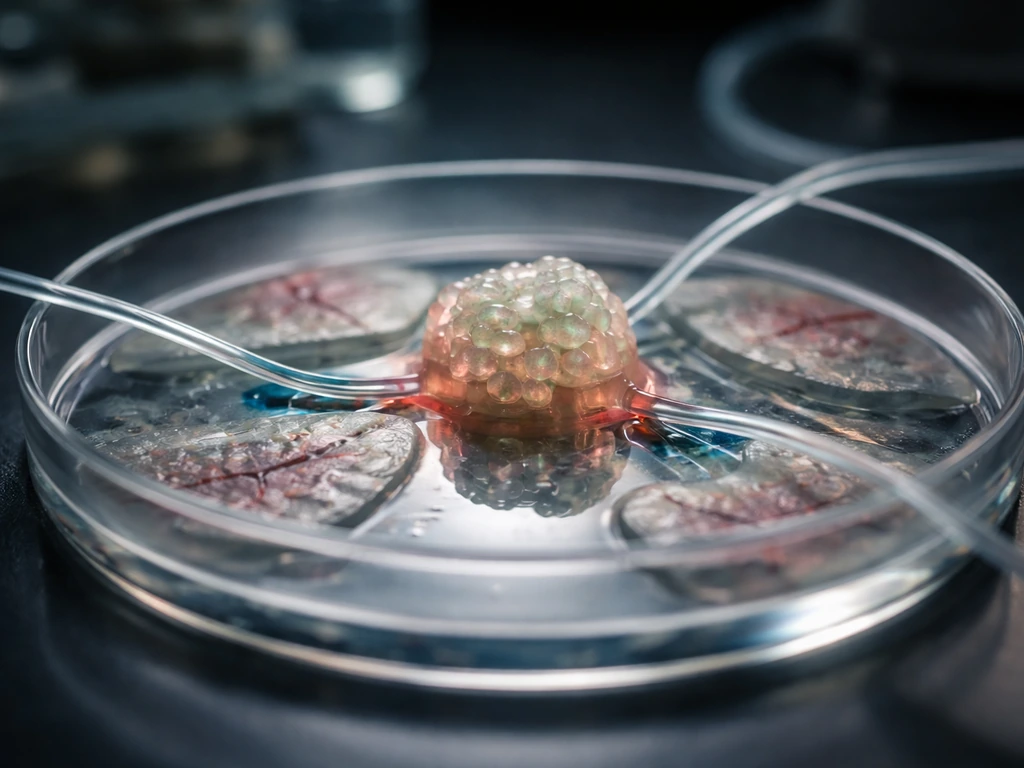
What 'growing organs' actually means today

The phrase 'grow an organ' covers a surprisingly wide range of things, and it matters a lot which one you're talking about. At one end of the spectrum, you have organoids: 3D structures grown from stem cells in a dish that self-organize into something resembling an organ's cell types and architecture. The International Society for Stem Cell Research (ISSCR) defines an organoid as a tissue-culture-derived 3D structure that recapitulates the cell composition and a subset of the physiological functions of an organ through self-organization. The key word is 'subset.' An organoid is a model, not a replacement.
Further along the spectrum, you have bioengineered tissue patches: lab-grown cartilage, skin, or corneal tissue that can actually be transplanted into patients to repair a specific defect. These are clinically real and already happening. At the far end, the goal most people imagine when they search 'can we grow organs,' is a complete, transplant-ready organ with full vasculature, innervation, and immune compatibility. That's still largely in the future, though the research moving toward it is genuinely exciting.
There's also a fourth category worth knowing: organ-on-a-chip systems, which use microfluidic devices to mimic organ function for drug testing. They're powerful tools, but even more stripped-down than organoids in terms of physiological complexity. To understand what scientists can actually grow in the lab, you need to hold all four categories in your head at once, because progress is happening at every level, just at very different speeds.
What's been achieved right now
The wins are real, and they're worth celebrating. Engineered cartilage is arguably the most clinically advanced example. CARTISTEM, a product using umbilical cord blood-derived mesenchymal stem cells combined with sodium hyaluronate, has been tested in Phase I/IIa clinical trials for full-thickness articular cartilage defects in the knee. Separately, programs using autologous dermis-isolated adult stem cells (DIAS cells) are working their way through preclinical-to-clinical translation for cartilage repair. These aren't science fiction; they're registered trials with defined endpoints.
On the organoid side, one of the most striking recent milestones was a first-in-human trial transplanting iPSC-derived retinal organoid grafts into patients with retinitis pigmentosa. Retinal tissue is relatively thin and avascular, which makes it a more tractable early target than, say, a liver. The fact that it crossed into human trials at all is a genuine landmark. Understanding how stem cells contribute to organ growth helps explain why these retinal grafts are possible now while kidney or heart replacements are further off.
NIH has also recently launched the Standardized Organoid Modeling (SOM) Center, with initial focus on standardized organoid models of liver, lung, heart, and intestine. This is significant not just as a research milestone but as an acknowledgment that reproducibility and assay standardization are now real bottlenecks, not only discovery. The field has matured enough that the problem has shifted from 'can we make organoids at all' to 'can we make them the same way twice, reliably.'
Why growing organs is genuinely hard: biology first
To understand why organ growth is so difficult to engineer, you need to understand how it works in nature. When a human embryo develops an organ, it's not just a pile of cells dividing until they reach the right size. It's a highly choreographed process driven by cell differentiation, self-organization, mechanical forces, and chemical signaling that happens across precise windows of time.
Differentiation and self-organization

Every cell in your body carries the same DNA, yet liver cells look and behave nothing like neurons. That transformation, called differentiation, is guided by morphogens: signaling molecules that diffuse through tissue and create concentration gradients. Cells read where they are in the gradient and decide what type of cell to become. This is the positional information concept, and it's one of the foundational ideas in developmental biology. Turing-style reaction-diffusion mechanisms further sharpen these patterns, creating the kind of precise spatial organization that complex organs require. Reproducing these gradients artificially, in the right sequence, at the right scale, is enormously difficult.
To understand how cells in our organs grow and coordinate their behavior, you have to appreciate that growth and differentiation are tightly coupled. Cells don't just divide and then decide what to become; the mechanical and chemical signals they receive during division actively shape what they turn into.
The extracellular matrix: more than scaffolding
One thing that often gets underappreciated outside of biology is the role of the extracellular matrix (ECM). The ECM isn't just a passive scaffold holding cells in place. It's an active signaling environment. Cells sense mechanical signals from the ECM through a process called mechanotransduction, and those signals influence cell fate, proliferation, and tissue morphogenesis. The stiffness of the matrix, its fiber orientation, its biochemical composition: all of it matters. Replicating the right ECM environment in a lab-grown structure is a major engineering challenge. Tuning ECM and mechanics is increasingly recognized as a key platform capability for organ engineering, not just a detail.
The physical constraints that stop organs from just 'growing bigger'

Here's a problem that any student of biology will find familiar: cells need oxygen and nutrients, and those things can only travel so far by passive diffusion. In living tissue, the limit is roughly 100 to 200 micrometers from the nearest blood vessel. Beyond that distance, cells start to starve and die. This is the same principle that limits how large a single cell can grow before its surface-area-to-volume ratio becomes a problem, just scaled up.
In statically cultured engineered cartilage constructs, researchers have measured that the core regions develop microenvironments with very low oxygen and nutrient levels, leading to cell death. Spinner flask culture systems can improve penetration, but nutrient transport depth in larger scaffolds can still be limited to around 1 mm. Perfusion bioreactor systems, which actively pump media through constructs, improve cell viability and nutrient exchange significantly, but they introduce their own engineering complexity, including shear stress on cells.
In living tissue, this diffusion problem is solved by an intricate network of blood vessels that reaches almost every cell in the body. Without that network, any lab-grown tissue thicker than a few hundred micrometers will have a dead core. This is the vascularization bottleneck, and it's probably the single biggest physical constraint on organ growth today.
The biggest remaining bottlenecks
Several technical walls still stand between current organoid science and transplant-ready organs. These aren't minor gaps; they're deep engineering and biology problems that entire research programs are dedicated to solving.
| Bottleneck | Why it matters | Current status |
|---|---|---|
| Vascularization | Without blood vessels, tissue beyond ~200 μm from a vessel dies; organs need dense capillary networks | Vascularized organoids-on-chip with demonstrated anastomosis and perfusion exist; host integration in vivo remains unsolved at scale |
| Maturation | Lab-grown cells often resemble fetal rather than adult tissue; immature cells may not perform adult organ functions | Microfluidic and perfusion platforms show improvement; still a major gap for most organ types |
| Immune compatibility | Transplanted tissue triggers immune rejection; iPSC-derived organoids express HLA class I antigens | HLA knockout and immune-evasion strategies are in development; no universal solution yet |
| Architecture and integration | Organs have precise 3D architecture and must connect to host vasculature, nerves, and ducts | Scaffold and bioprinting approaches advancing; host integration after transplant poorly understood |
| Reproducibility and scale | Clinical use requires consistent, scalable manufacturing | NIH's SOM Center now directly targeting standardization for liver, lung, heart, and intestine |
Vascularization deserves special attention. NIH has explicitly noted that a key limitation of standard organoids is that they 'lack a blood vessel system,' and that overcoming size limits requires solving this. Recent work has shown that microfluidic platforms can integrate functional vascularized organoids-on-chip with demonstrated anastomosis and controlled perfusion, which are stronger functional indicators than static tissue staining alone. But getting those vessels to connect to a host's circulation after transplant is a different and harder problem.
Immune compatibility is the other major wall. A study of T-cell mediated immune rejection in iPSC-derived kidney organoids found that these organoids express HLA class I antigens, making them vulnerable to rejection. Strategies like HLA knockout are being explored, but they introduce their own risks. This is why NIAID has a dedicated Notice of Special Interest targeting development of 3D organotypic culture models for transplantation immunology research, specifically to understand and address immune interactions before clinical translation.
One question that comes up naturally here is what happens to organs once they're actually transplanted: do transplanted organs continue to grow or remodel after they're placed in a recipient? The short answer is that they can adapt, but they don't grow to fill a size mismatch the way developmental tissue would. That distinction matters for understanding what 'integration' really means.
The complexity question: can we even grow a brain?
It's worth pausing on the most extreme example to calibrate expectations. Brain organoids, tiny self-organized structures that partially mimic cortical development, exist and are being used to study neurological disease. But the question of whether you can actually grow a functional brain in a lab exposes just how far the field has to go. Brain organoids lack the precise layered architecture, the vascular supply, the glial support networks, and the circuit-level organization of a real brain. They are profoundly useful research tools, but calling them 'brains' would be like calling a weather map a storm.
When might we actually see transplantable grown organs?

Honest timeline expectations require separating organ types by complexity. Thin, relatively avascular tissues (cornea, retina, skin) are closest. Cartilage and bone are next. Solid vascularized organs (liver, kidney, heart, lung) are furthest. Here's a rough framework based on current trajectories:
| Organ type | Current stage | Realistic near-term outlook |
|---|---|---|
| Retinal tissue | First-in-human trials completed | Incremental clinical refinement over next 5–10 years |
| Cartilage | Phase I/IIa clinical trials; multiple programs | Expanded clinical use plausible within a decade |
| Skin grafts | Already in clinical use (burns, wounds) | Continued improvement in complexity and functionality |
| Liver organoids | Preclinical; NIH SOM standardization underway | Partial replacement or assist devices possible in 10–20 years; full replacement much longer |
| Kidney | Organoids exist; immune rejection and vascularization unsolved | 20+ years to transplant-ready with current trajectory |
| Heart | Patches and partial constructs in early trials; full organ not feasible | Partial patches advancing; whole heart replacement decades away |
The milestones that matter most to watch are: first demonstrated host vascularization of a lab-grown solid organ in a large animal model, successful long-term immune tolerance of an iPSC-derived graft without systemic immunosuppression, and standardized manufacturing protocols that produce consistent organoids across labs. The FDA's regulatory framework for regenerative medicine products already anticipates this pipeline, using risk- and quality-focused approaches rather than treating all grown tissues as a single category, which matters because it means regulators are thinking ahead about how to evaluate these products when they arrive.
There are also broader questions running alongside the science. Debates about whether scientists should be allowed to grow animals in artificial wombs for organ sourcing touch on some of the same ethical territory, even if the technology differs. These conversations are happening in parallel with the lab work, not after it.
The ethics aren't separate from the science
Growing organs isn't just a technical challenge. The question of whether it's ethical to grow human organs at all, especially using stem cells, chimeric approaches, or genetic modification, is genuinely contested. ISSCR guidelines exist specifically to provide oversight and translation principles for evaluating whether claimed 'organs' are clinically ready versus still experimental model systems, and to ensure that ethical review keeps pace with technical capability. This matters practically: it shapes which research gets funded, which trials get approved, and how fast the field can move.
What you can actually do with this knowledge today
If you're a student, educator, or curious learner trying to track this field, here's how to stay oriented and evaluate what you read:
- Distinguish the category. When you see a headline about 'grown organs,' ask: is this an organoid (a 3D model), a tissue patch (transplantable but limited), or a full organ claim? Most claims are the first two. Full organ claims at clinical scale are not yet real.
- Check for vascularization evidence. A meaningful claim about a grown organ should show functional vasculature, not just labeled staining. Look for terms like anastomosis, perfusion, or host integration, and be skeptical of size claims without vascular data.
- Verify the trial stage. ClinicalTrials.gov lets you look up any therapy by name or identifier. Check whether it's Phase I (safety only), Phase II (early efficacy), or Phase III (large-scale). CARTISTEM-level Phase I/IIa data is genuinely promising. A press release without a trial number is much weaker evidence.
- Follow NIH's SOM Center outputs. This center is specifically standardizing organoid models for liver, lung, heart, and intestine. Their publications and updates are a reliable signal of where reproducibility and translation milestones are being hit.
- Track immune compatibility research. NIAID's active funding priority for transplantation immunology in 3D culture models tells you that immune rejection in organoid-derived grafts is still a major unsolved problem. Any therapy claiming to have solved this deserves careful scrutiny.
- Learn the underlying biology. Understanding cell division, differentiation, ECM signaling, and diffusion constraints gives you a framework to evaluate any new claim. These aren't just academic details; they're the actual reason organ growth is hard.
- Read peer-reviewed sources for landmark claims. The retinal organoid first-in-human trial was published in Cell Stem Cell with defined clinical endpoints. That's a very different evidence level from a university press release. Use PubMed to check whether a claim has that kind of backing.
One area worth dedicating real learning time to is how scientists cultivate enough cells to even begin building these structures. How scientists grow enough cells for their research is a genuine bottleneck question, because you need billions of correctly differentiated cells to build any meaningful tissue structure, and scaling that process without losing cell quality is harder than it sounds.
The honest bottom line
We can grow organs in the sense that we can produce organoids, engineer tissue patches, and even transplant some of these into human patients. We cannot yet grow a full-sized, vascularized, innervated, immune-compatible organ ready for transplant off the shelf. The gap between those two things is real, but it's closing. The biological mechanisms that make organ growth hard, diffusion limits, differentiation complexity, ECM signaling, immune recognition, are all being actively worked on, and for the first time, standardization and reproducibility are being treated as first-class problems rather than afterthoughts. That's a sign of a maturing field, not a stalled one.
FAQ
If researchers can transplant lab-grown tissue, does that mean we can already grow full organs for replacement?
Not for most current approaches. “Organoid” and “patch” therapies usually start with a specific tissue defect and aim for functional repair, not full organ replacement. For a true replacement (kidney, liver, heart), the biggest missing elements are rapid, durable host-level vascular connection and immune acceptance throughout the entire tissue, not just in a small graft area.
How can I tell whether a “grown organ” claim is actually an organoid, a patch, or a transplant-ready replacement?
Often no, because “organs” in headlines can mean different things. A practical rule is to ask whether the product is a thin cell layer or model (research), a localized patch (repair), or a solid transplant candidate (replacement). If it cannot connect to host blood supply and sustain function over time, it is still closer to a model or patch than a full organ.
Will someone who receives a lab-grown graft always need immunosuppression?
Care plans can differ dramatically. Many early graft approaches still require tailored immunosuppression or specific strategies depending on the cell source and the immune compatibility profile. Even if studies reduce rejection risk, protocols may involve monitoring HLA-related responses and graft behavior rather than assuming the body will accept the tissue automatically.
Why do organ-like tissues fail when they are made larger, even if the cells are correct?
Because diffusion limits scale with tissue thickness, many constructs are designed with controlled geometry. Even small changes in scaffold architecture can affect oxygen and nutrient penetration. If a graft is made thicker or denser without improved perfusion, the “dead core” problem can appear even when the cells themselves are healthy.
Do transplanted organoids or grafts grow to fill the missing size of an organ?
Yes, but it is not usually the same as embryo-style growth. After transplantation, tissues can remodel and integrate locally, but they typically do not expand to match an adult organ’s exact size and complex structure. Integration means survival and functional incorporation in a host environment, not re-running full developmental programs to fill a size mismatch.
Why are retina, cornea, and cartilage often ahead of liver or kidney in progress?
It depends on the construct. Some lab-grown tissues are designed to be relatively avascular (for example, thin tissues), making early targets more feasible. For thicker, solid organs, success increasingly depends on vascularization strategies such as perfused microvascular structures or platforms that support vessel function before and after transplant.
What makes “replicating the same organoid twice” so difficult, and why does it matter for patients?
Yes. The most common manufacturing issue is variability between batches, which can change cell maturity, composition, and functional readouts. Standardization efforts focus on making organoid generation reproducible across labs and over time, using defined assays and quality checks rather than relying on appearance alone.
How long does it typically take from lab-grown tissue success to actual clinical availability?
Not immediately, and not in a one-size-fits-all way. Researchers have to prove safety and function for each product type and indication, especially around immune responses, tumor risk from stem-cell derivatives, and long-term graft behavior. Regulatory pathways for regenerative products are designed for risk and quality controls, so translation is usually slower than the science headlines suggest.
What are common misconceptions about how “vascularized” organoids are in research papers?
Yes, and it can be counterproductive if it hides the real constraints. Be cautious with claims that tissue is “vascularized” based only on staining, or that vessels will automatically anastomose and connect to host circulation. Stronger evidence involves demonstrating perfusion and functional integration, not just the presence of vessel-like structures.
Why are brain organoids described as models instead of real brains, even though they develop in 3D?
Not always. Even when a tissue looks like the target organ under a microscope, it can lack key components needed for real-world function, such as correct layered organization for brain-like tissues, sufficient support networks, or circuit-level activity. That is why many researchers emphasize models and functional assays rather than using organ labels loosely.
How can organoids or organ-on-a-chip systems help, if they cannot replace organs yet?
A better expectation for research impact is usually “faster and safer development,” not instant replacement. Organs-on-chips and organoids can reduce reliance on animal studies for drug testing and help identify toxicity earlier, but they are not full substitutes for whole-organ physiology and immune interactions.
Do Transplanted Organs Grow? What Actually Happens After Transplant
Do transplanted organs grow? Learn how they adapt via hypertrophy and remodeling, why limits apply, and how it’s monitor