Paramecium grow in two distinct ways: an individual cell increases in size as it feeds and prepares for division, and the population grows as cells split into two through binary fission. Both processes depend on the right conditions, and if either breaks down, growth stalls. Here is exactly how it works and what you can do to keep a culture thriving.
How Do Paramecium Grow: Growth Cycle, Conditions, and Tips
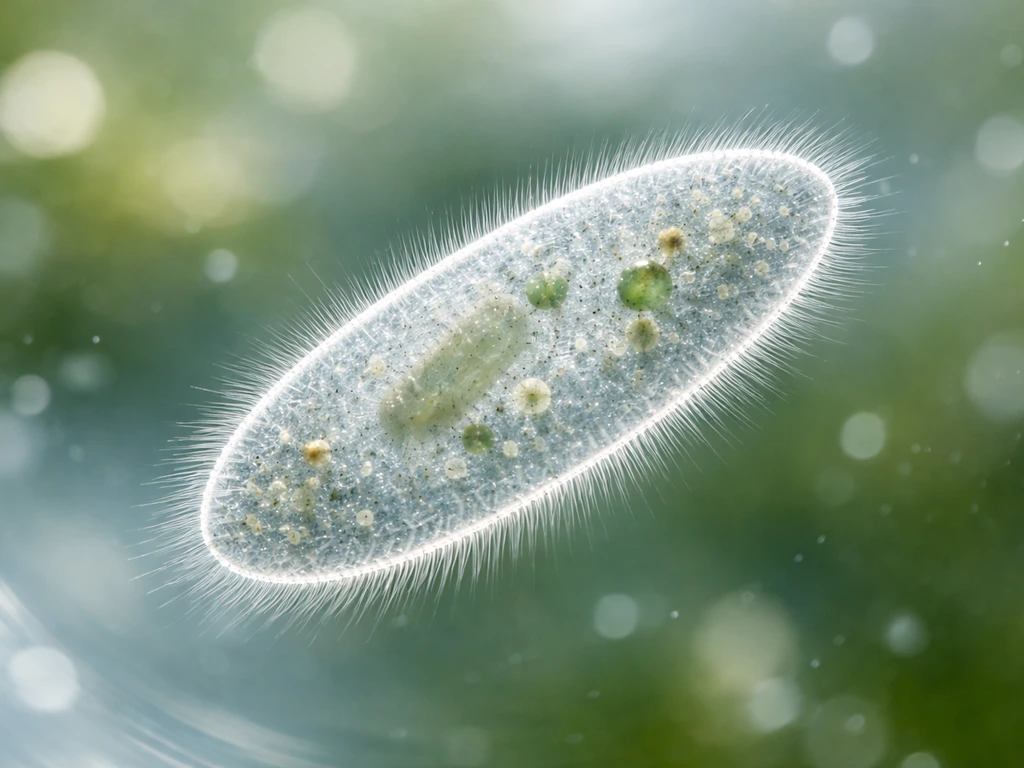
What 'growth' actually means for a single-celled organism

Paramecium are ciliates, single-celled protists covered in rows of tiny hair-like cilia that beat in coordinated waves to propel the cell and sweep food into its oral groove. When most people ask how Paramecium grow, they are really asking two questions at once. First, how does one cell get bigger? Second, how does a culture go from a few cells to thousands? The answers are related but not the same thing.
For a single-celled organism, 'growth' means increasing cell mass, synthesizing new proteins and organelles, and eventually reaching a size threshold that triggers division. There is no childhood, no adolescence, and no adult phase the way we think of in multicellular animals. The cell cycle is the whole story. Understanding this is the key to understanding why conditions like food availability and temperature affect growth so dramatically, and it connects directly to how protists grow and develop more broadly.
Feeding, getting bigger, and the cell cycle
Growth starts with eating. Paramecium feed primarily on bacteria and small organic particles. Cilia lining the oral groove funnel food toward the cytostome (the cell's 'mouth'), where particles are engulfed into food vacuoles at the cytopharynx. Those vacuoles then circulate through the cytoplasm in a predictable route, fusing with lysosomes that release digestive enzymes. Nutrients are absorbed, and waste products are eventually expelled at the cytoproct, a fixed exit point on the cell surface. This continuous ingestion-and-digestion loop supplies the raw materials the cell needs to synthesize more of itself.
As nutrients accumulate, the cell grows physically larger. Ribosomes crank out new proteins, the cytoplasm expands, and organelles like mitochondria replicate. The macronucleus, which handles the day-to-day gene expression that drives vegetative functions, coordinates this growth phase. The micronucleus, meanwhile, holds the germline genetic material and becomes critically important during sexual reproduction. Both nuclei must eventually divide before the cell can split.
Think of the cell cycle as a build-then-divide rhythm. There is a growth phase where the cell accumulates mass and duplicates its contents, and then a division phase where everything splits. Under optimal conditions, Paramecium caudatum can complete this cycle up to three times within a single 24-hour period, meaning one cell can theoretically become eight cells in a day. That is fast, and it explains why a well-fed culture in the right temperature range can seem to explode in numbers almost overnight.
Binary fission: how one Paramecium becomes two
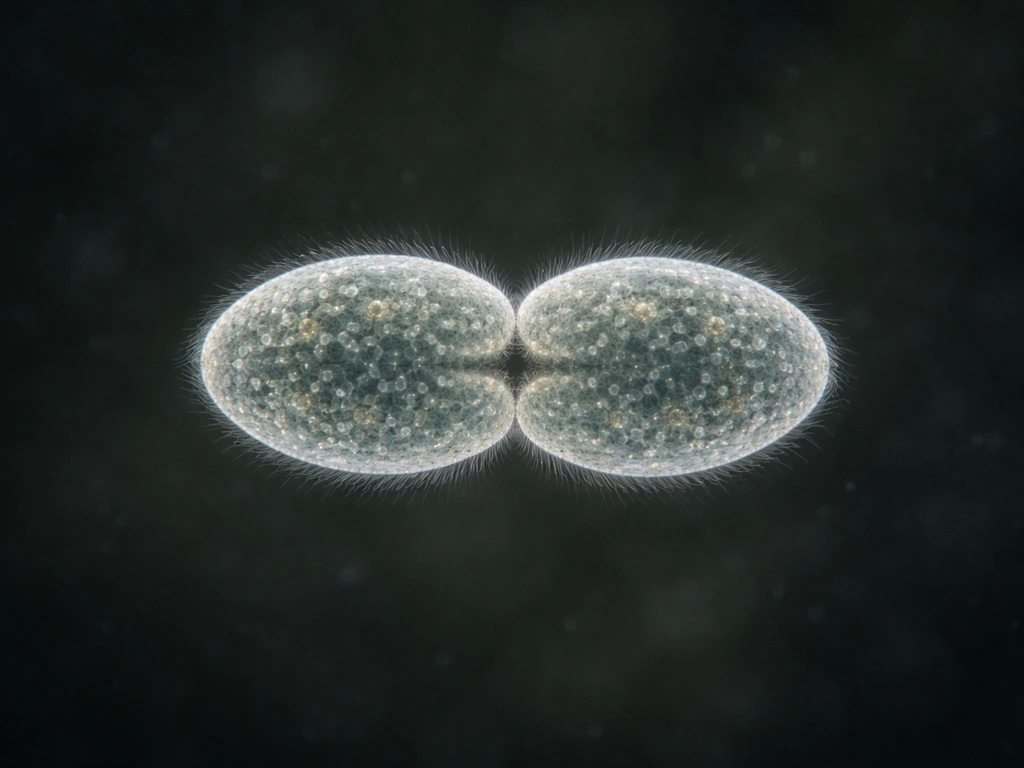
The primary way Paramecium reproduce is transverse binary fission, which is asexual division across the short axis of the cell. The process is directed by the macronucleus. During fission, a constriction furrow forms roughly in the middle of the cell and deepens until the two daughter cells pinch apart. As this happens, oral structures like the buccal cavity can actually disappear and then reform in each daughter cell. Each daughter inherits roughly half the original cytoplasm, organelles, and nuclei, then immediately begins feeding and growing toward its next division.
Paramecium also has a sexual process called conjugation, where two compatible cells pair up, exchange micronuclear material, and rebuild their macronuclei. This is not reproduction in the sense of making more cells, but it resets the genetic slate and is important for long-term culture health. The conjugation process is elaborate: micronuclei go through meiosis and mitotic divisions, the original macronucleus fragments and degenerates, and development of the new macronucleus takes roughly 85 of the approximately 110 hours the whole process requires. In practice, you are more focused on binary fission when trying to grow a culture, but conjugation matters for maintaining genetic vitality over many generations.
This pattern of asexual division as the core growth mechanism is worth comparing to how other microorganisms expand. Amoeba grow through a similar build-and-divide logic, though their feeding and movement strategies differ considerably from the ciliate approach Paramecium uses.
The conditions Paramecium need to actually grow
Temperature
Temperature is one of the most important levers you have. Optimal growth for Paramecium caudatum generally falls between 24°C and 28°C (roughly 75°F to 82°F). One lab facility protocol specifies maintaining cultures at 24 to 26°C (75 to 79°F) and explicitly warns against temperature fluctuations, since constant conditions are what cultures grow best in. Research confirms the effect: daily fission rate at 24°C is measurably lower than at 27°C, so even a few degrees makes a real difference in how fast your culture builds up. Below about 20°C, division slows significantly. Above 30°C, heat stress kicks in and cells start dying faster than they reproduce.
Food availability

You cannot separate growth from feeding. In most lab and classroom cultures, Paramecium are fed bacteria that grow on an organic medium: boiled wheat grains, lettuce infusion, or dried hay are common substrates that cultivate the bacterial food source. The bacteria do the work of converting organic matter into a form Paramecium can eat. If the bacterial population crashes, or if you add food medium that is too rich and triggers a crash in water quality, Paramecium growth stalls. Balance is everything.
Water quality
Paramecium are sensitive to water chemistry. They do best in dechlorinated, clean freshwater. Some lab protocols filter the water used for growing cultures through a 0.23 µm stainless-steel pressure filter before use. Tap water with chlorine or heavy chloramine treatment will kill or inhibit them. pH matters too: a range close to neutral (around 6.5 to 7.5) generally supports healthy growth. High ammonia from waste buildup or overfeeding will poison the culture. This is one reason partial water changes are a useful tool when managing a culture.
Oxygen and gas exchange
Paramecium are aerobic. They need dissolved oxygen to survive and grow. In a dense, rapidly growing culture, oxygen depletion can become a real problem. One practical recommendation is to aerate the culture periodically using a pipette tip to gently bubble air through the medium if you suspect oxygen is getting low. You do not need a fancy aquarium pump for small cultures, but you do need to make sure the container is not sealed airtight and that there is regular gas exchange at the surface.
Why growth cannot go on forever
Every Paramecium culture has a ceiling, and that ceiling is determined by the carrying capacity of the environment. Studies on Paramecium caudatum found an average maximum density of around 300 cells per mL under controlled conditions, though the number varies by clone and container setup, with ranges reported anywhere from about 70 to 600 cells per mL depending on conditions and genetic strain. Once a population approaches carrying capacity, several factors push back simultaneously.
- Food runs out faster than it is replenished, starving cells before they can complete a full growth cycle.
- Metabolic waste, including CO₂ and ammonia compounds, accumulates and becomes toxic at high concentrations.
- Physical crowding can trigger density-dependent behavioral responses, including dispersal, where cells actively move away from overcrowded areas.
- The fission rate slows as energy budgets tighten, and cell death begins to balance or exceed new divisions.
This concept of resource limits capping growth is not unique to Paramecium. It plays out across microscopic life in similar ways, and it is one reason plankton growth in natural water bodies follows similar boom-and-bust patterns tied to nutrient availability and light.
There is also a cellular-level constraint worth knowing about. Paramecium, like all cells, face physical limits on how large a single cell can get before diffusion becomes inefficient. Oxygen, nutrients, and signaling molecules have to reach the interior of the cell, and the surface-area-to-volume ratio shrinks as a cell gets bigger. This is a universal ceiling on cell size, not just a Paramecium quirk. It is why division is the only viable long-term growth strategy for a single-celled organism. The same logic applies to organisms that seem very different, like the colonial structures formed by diatoms as they grow, where individual cell size is also tightly constrained.
How to observe and measure growth in your culture
Seeing what healthy looks like
Healthy Paramecium are actively swimming in rapid, spiral patterns. Under a microscope at around 40× with darkfield illumination you can spot them moving across the field; bumping up to 100× lets you watch feeding and behavioral responses; and 400× phase contrast reveals the cilia themselves and the rotating motion of food vacuoles inside the cell. A healthy cell is elongated and slipper-shaped, roughly 50 to 300 micrometers long depending on species, with uniform ciliation and visible oral groove activity. Stressed or dying cells move sluggishly, clump together, or appear swollen and irregular.
Counting and measuring population growth

The most reliable way to track culture growth is by counting cells using a hemocytometer. Load a small sample into the chamber, count cells across consistent grid squares, and apply the standard conversion: cells per mL equals the mean count per square multiplied by 10,000 (10^4). Always apply a dilution factor correction if you diluted the sample before loading. Consistency matters: use the same grid squares every time and apply the same counting rules for cells sitting on boundary lines. Repeat measurements every 24 to 48 hours and plot the results to see whether your culture is in a lag phase, exponential phase, or approaching its carrying capacity.
If a hemocytometer is not available, you can get a rough relative count using a glass well slide and a hand tally counter under a binocular microscope, counting cells in a defined volume of sample. It is less precise, but it will tell you whether numbers are going up, staying flat, or crashing.
Troubleshooting when your culture stops growing
Growth stalls for predictable reasons. Here is how to diagnose and fix the most common problems.
| Problem | Likely Cause | Fix |
|---|---|---|
| Cells moving slowly or clumping | Oxygen depletion or temperature too low | Aerate with a pipette tip; move culture to 24–27°C |
| Population dropping rapidly | Food source crashed or water quality poor | Refresh culture medium; do a 50% water change with dechlorinated water |
| No growth after a week | Water too cold, chlorinated, or contaminated | Check temperature (aim for 24–27°C); use filtered or aged dechlorinated water |
| Culture smells foul | Bacterial overgrowth or overfeeding | Reduce food input; transfer a small aliquot to fresh medium to restart |
| Cells appear swollen or bursting | Osmotic stress or toxin exposure | Check water source; avoid tap water with chloramine; use fresh filtered water |
| Density plateaus below 100 cells/mL | Underfeeding or low bacterial density | Boost food medium; confirm bacteria are present and thriving in the substrate |
One thing that catches beginners off guard: adding too much food at once is just as dangerous as too little. A heavy dose of organic matter fuels a bacterial explosion, which then crashes and rots, dropping oxygen and spiking ammonia. Add food in small, regular amounts rather than large infrequent dumps.
If a culture has completely collapsed and smells terrible, do not try to rescue it directly. Instead, use a small starter sample (even 1 to 2 mL) transferred to completely fresh medium. Paramecium cultures can bounce back from very low densities if conditions are corrected. This is also why keeping a backup culture in a separate container is good practice for anyone running paramecia long-term.
Putting it all together: a practical growth checklist
- Maintain water temperature at 24–27°C (75–80°F) with minimal fluctuation.
- Use dechlorinated, filtered water (0.23 µm filter if possible, or age tap water for 48 hours).
- Feed with a bacterial culture on boiled wheat grains, hay, or lettuce infusion — add small amounts every few days, not large dumps.
- Keep containers open or loosely covered to allow gas exchange; aerate if you notice sluggish movement.
- Count cells every 1 to 2 days using a hemocytometer or well slide to track whether the culture is growing, plateauing, or crashing.
- Do partial water changes (about 30–50%) every 5 to 7 days to remove waste and prevent ammonia buildup.
- Watch for behavioral signs: fast spiral swimming means healthy; slow or clumped movement means something is wrong.
- Keep a backup culture in a separate container to rescue from if the primary crashes.
Paramecium growth is really a story about conditions. Get the temperature, water quality, food, and oxygen right, and binary fission does the rest. The biology is elegant, and once you have seen exponential growth in a culture you managed yourself, it is hard not to find the whole thing fascinating. For anyone curious about how this kind of growth compares across other microbial life, looking at how phytoplankton grow offers a useful contrast, since photosynthetic microorganisms add light as a major variable that ciliates do not depend on. And if you want to think about what growth even means for entities that blur the line between living and non-living, the question of whether viruses grow and develop is a genuinely interesting rabbit hole. The one thing all of these cases share: growth is always constrained, always conditional, and always more interesting when you look closely at the mechanisms behind it.
Understanding those constraints is also what separates a thriving culture from a dead one. Paramecium are not fragile if you respect what they need, but they will not grow on goodwill alone. Stick to the checklist, watch your cells, and adjust early when something looks off. That is the whole game. It also connects naturally to thinking about how parasites grow inside host environments where resources and conditions are similarly constrained but in much more dynamic and adversarial ways.
FAQ
Do Paramecium grow by getting bigger first, or do they immediately start dividing?
They do not increase cell size indefinitely. A single Paramecium grows for part of its cell cycle, then reaches a threshold where division is triggered. If you keep feeding and conditions are good, you will mainly see more cells produced rather than one cell becoming dramatically larger over time.
How can I tell if growth is limited by food for the Paramecium or by the bacteria they eat?
A sharp slowdown usually comes from the “food chain,” not from the Paramecium directly. If bacteria crash due to overfeeding, poor medium prep, or waste buildup, Paramecium will stall even if the culture still looks like it has liquid nutrients.
My culture was growing, then suddenly slowed down. What should I check first?
Most common causes are oxygen shortage, water chemistry drift (ammonia or pH shift), and temperature stress. The fastest indicator is behavior: stressed cells typically slow down, clump, or look swollen. If you see that, correct temperature first, then check aeration and consider a partial water change rather than adding more food.
Will adding more bacteria always make a Paramecium culture grow faster?
Overfeeding can create a bacterial boom followed by rotting, which raises ammonia and reduces oxygen. Instead of adding “more to speed things up,” feed in smaller, consistent portions and only increase feeding if you confirm numbers are still rising and waste smells are not getting worse.
What is the best rescue strategy if my culture crashes or smells strongly?
If the culture is collapsing, the safest approach is to transfer a small amount into fresh, matched medium (same temperature range, dechlorinated water, and similar pH). Because Paramecium tolerate low densities better than they tolerate ongoing chemical stress, a reset often works even after near-zero growth.
Can I grow Paramecium in a closed or tightly sealed container?
Paramecium are not ideal for sealed containers because they need gas exchange. Even if oxygen is present initially, a sealed setup limits replenishment and can trigger later oxygen depletion during rapid growth.
Why does carrying capacity seem to differ from one culture to another?
Yes, and it changes growth behavior. Different strains and setups can reach different maximum densities, and higher densities also make oxygen depletion and waste buildup more likely. If you want faster growth, aim for exponential-phase numbers rather than trying to push toward the absolute maximum density.
At what point should I thin or transfer a culture instead of letting it keep multiplying?
Yes. If a culture is too dense, oxygen drops and waste builds up faster than feeding can offset it, which can flip growth from exponential to flat or declining. Regular monitoring with cell counts helps you decide when to thin, transfer, or refresh medium.
What are the most common mistakes when counting Paramecium with a hemocytometer?
Use counting consistency and dilution discipline. Count the same hemocytometer grid each time, apply the dilution factor if you diluted the sample, and treat cells on boundary lines using a consistent rule (for example, include those touching the top and left edges only). This prevents “phantom growth” from counting variation.
If I do not have a hemocytometer, how can I still track whether my culture is in lag, exponential, or decline?
If you do not have a hemocytometer, relative counts can still track trends, but you should define a repeatable sampling volume and count time. Use the same slide type, magnification, and scanning pattern each session so you are comparing similar portions of the culture.
Does conjugation increase the number of Paramecium in my culture?
Short answer, conjugation usually does not increase cell numbers. It rebuilds genetic material and helps long-term vigor, so if your goal is rapid culture expansion you mainly rely on asexual division, while conjugation is more about resetting health when needed.
How Does Phytoplankton Grow? Steps, Limits, and Conditions
How phytoplankton grow: photosynthesis, nutrient uptake, cell division, and the limits set by light, heat, nutrients, mi


