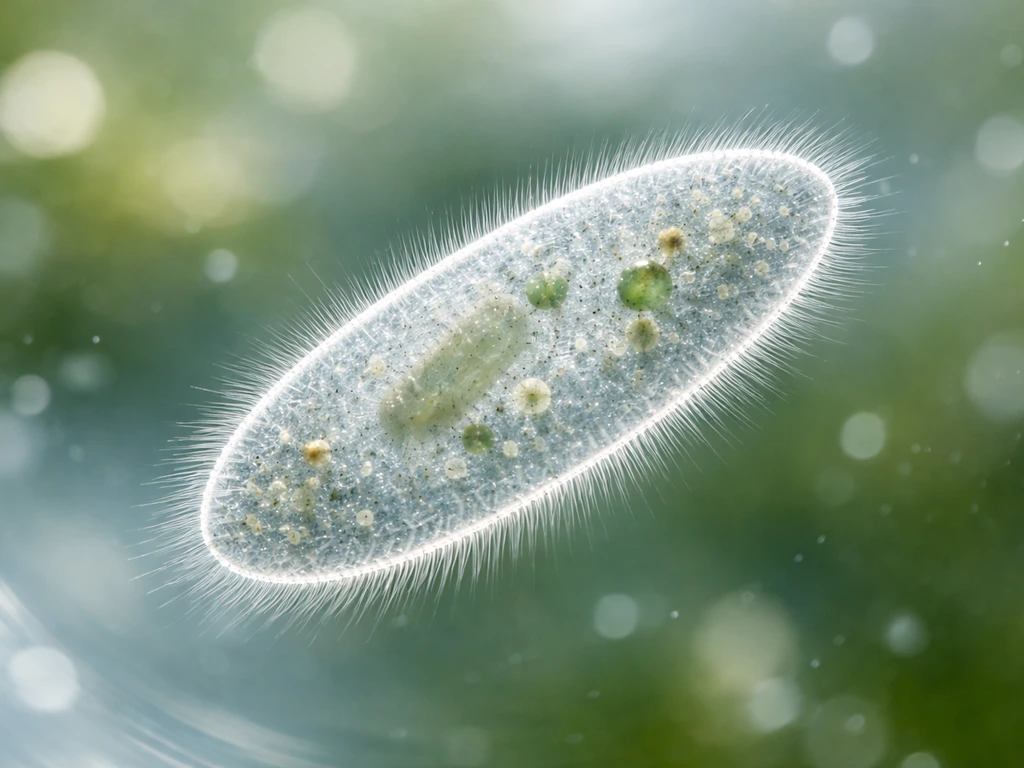
Protists grow by doing two things at once: individual cells get bigger by accumulating biomass, and then they divide to produce more cells. Development, in protists, doesn't mean building organs or tissues. It means shifting between life-cycle stages, like going from an active feeding cell to a dormant cyst and back again. These stage changes are part of how plankton grow and shift between growth and dormant forms. Both processes are tightly controlled by nutrients, light, temperature, and a handful of physical laws that apply to every living cell on Earth.
How Do Protists Grow and Develop: Cell Growth and Division

What 'growth and development' actually mean for a protist
It helps to separate these two ideas before mixing them together. Growth, in the biological sense, means an increase in biomass, either within a single cell or across a population of cells. Development means a change in form or function over time. For animals, development conjures images of embryos and organs. For protists, it's more subtle but still real.
Many protists are permanently unicellular, yet they still show differentiation of cellular morphology and function. A cell preparing to divide looks and behaves differently from a cell that just finished dividing. A cell under nutrient stress may dramatically reorganize its internal architecture. Some protists even form loose colonial structures, where cells specialize slightly without becoming truly multicellular. So 'development' in protists is better understood as stage-based change rather than tissue-based change.
This distinction matters practically. If you're watching a protist culture in a lab, you're not just counting cells. You're watching cells cycle through phases of growth, division, and sometimes dramatic structural transitions, all without a single muscle cell or neuron in sight.
How a protist cell actually gets bigger
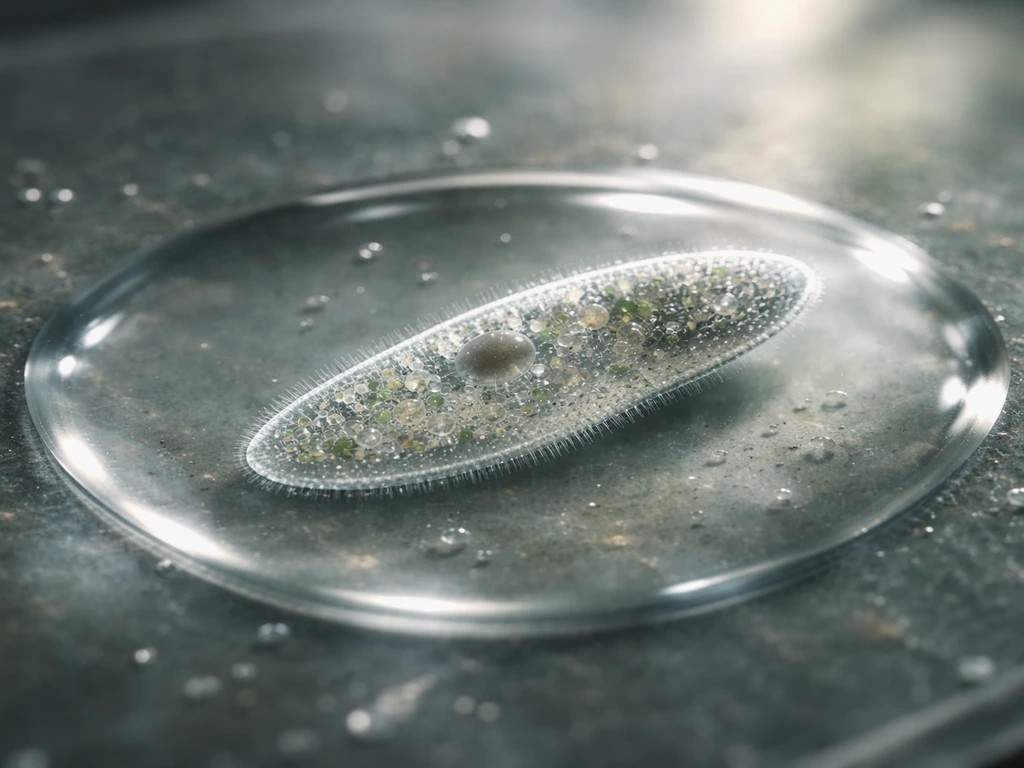
Before a protist can divide, it has to grow. Paramecium also needs to take in enough nutrients and convert them into new cell components before it can divide and increase in number. Photosynthetic plankton and other phytoplankton follow the same basic pattern of nutrient-driven biomass buildup before they divide phytoplankton grow. That means pulling in raw materials from the environment and converting them into cellular machinery: proteins, membranes, organelles, nucleic acids. Photosynthetic protists like dinoflagellates and diatoms do this using light energy and dissolved CO2. Diatoms rely on light and dissolved CO2 to drive the photosynthetic chemistry that supports their growth Photosynthetic protists like dinoflagellates and diatoms. Heterotrophic protists, like amoebae and many flagellates, do it by engulfing bacteria or other food particles and digesting them for parts.
The rate at which a cell accumulates biomass depends directly on how much nutrient it has access to internally. Researchers who study algae and protists use something called the Droop quota model to capture this. The idea is straightforward: a cell's growth rate is a function of its internal nutrient quota (how much phosphorus, nitrogen, or another key element is stored per cell), not just what's floating around outside the cell. The relationship takes the form µ = µm'(1 − Q0/Q), where µ is growth rate, Q is the cell's internal quota, and Q0 is the minimum quota below which growth stops. In plain English: the more nutrient the cell has stored internally, the faster it grows, but there's a ceiling on how fast it can go, and a floor below which it simply can't divide.
Phosphorus limitation is one of the most commonly observed real-world constraints. Dissolved inorganic phosphorus can become depleted in both lab cultures and natural water bodies, and when it does, protist growth slows well before cells run out of carbon or light. The same principle applies to nitrogen. A cell that looks fine under a microscope may be internally starved and nearly growth-arrested.
Cell division: the engine that multiplies protist numbers
Once a protist cell has accumulated enough biomass, it divides. Because protists are eukaryotes, their division follows a eukaryotic mitotic framework: the nucleus replicates its DNA, chromosomes are segregated, and the cell splits into two daughter cells, each with a complete genome. That's the same core process operating in your own liver cells when they regenerate, just without all the surrounding tissue context.
But 'eukaryotic mitosis' covers a lot of variation. Dinoflagellates, for example, run an unusual form of closed mitosis where chromosomes associate with cytoplasmic spindle microtubules through membrane tunnels that pass directly through the intact nucleus. The nuclear envelope never breaks down the way it does in most eukaryotic cells. This is not a quirk but a deeply ingrained feature of dinoflagellate biology, and it shows how much variation exists within the broad category of 'protist cell division.'
Timing of division is also tightly regulated. Photosynthetic dinoflagellates grown on a 16:8 hour light-dark cycle enter S-phase (DNA replication) roughly 10 hours after the light comes on, and complete mitosis 14 to 16 hours after light onset. Cell division is essentially locked to the circadian cycle, happening at the end of the dark phase. This means light isn't just an energy source for photosynthetic protists. It's also a clock signal that gates when division is allowed to happen.
Life cycles and stage changes: development without any tissues
Protist life cycles range from dead simple (divide, divide, divide) to genuinely elaborate. Some parasitic protists have to infect multiple different host species at different developmental stages just to complete one full life cycle. This same growth logic also applies to parasitic life cycles, where parasites must reach the right developmental stage inside and between hosts parasitic protists. That's not random complexity. It's a highly coordinated developmental program running in a single-celled organism.
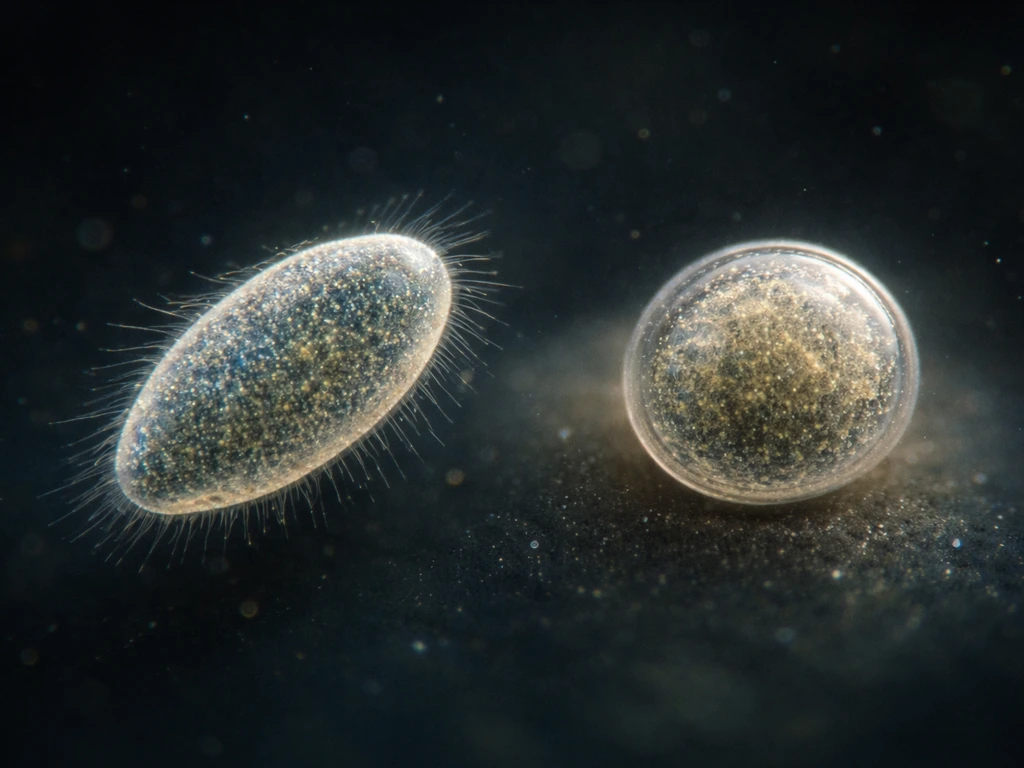
One of the most widespread developmental transitions in protists is encystment: the formation of a protective, dormant cyst. When conditions turn bad, whether from temperature stress, nutrient depletion, desiccation risk, or chemical signals, many flagellates, amoebae, and ciliates will trigger a cascade of internal changes. The cell loses complexity, sheds or retracts surface features, reorganizes or loses certain organelles, and deposits a resistant outer wall. The result is a cyst that can survive conditions that would kill the active cell outright.
This isn't passive or accidental. In ciliates like Colpoda cucullus, temperature-induced encystment involves multiple signaling pathways, with cAMP acting as a key second messenger that coordinates the transition. Microscopically, you can actually see the structural changes: ventral and dorsal features disappear, surface architecture shifts, and the cell shrinks and dehydrates. It's cell differentiation, just compressed into a single cell rather than spread across a developing embryo.
Excystation, the return to active growth when conditions improve, is equally regulated. For parasitic species like Entamoeba histolytica or Cryptosporidium, cyst formation is also a dispersal strategy. The cyst stage is what survives outside the host long enough to infect a new one. Understanding when and why protists encyst and excyst is genuinely important for anyone working with protist cultures, because an unexplained population crash may not be cell death. It may be mass encystment.
What controls how fast protists grow
Protist growth is not a single-dial system. Several environmental factors interact simultaneously, and knowing which one is actually limiting at a given moment is the key to predicting or managing growth.
| Factor | How it affects growth | Practical implication |
|---|---|---|
| Nutrients (P, N, trace elements) | Internal quota drives growth rate via Droop-type kinetics; depletion halts division before cells look visibly stressed | Monitor nutrient supply; don't assume cells look healthy means they're nutrient-replete |
| Light (for phototrophs) | Energy source AND clock signal; controls timing of cell division in circadian-regulated species | Match light cycle to species requirements; light duration affects when, not just if, cells divide |
| Temperature | Affects enzyme kinetics and can trigger encystment; growth optima vary widely across protist lineages | Stay within species-specific temperature range; sudden shifts can induce cyst formation |
| pH | High pH (above ~8.8) reduces CO2 availability and suppresses photosynthesis and growth rate | Buffer cultures appropriately; watch for pH drift in dense, photosynthetic cultures |
| Dissolved gases (CO2, O2) | CO2 is a carbon source for phototrophs; low O2 can shift metabolism toward anaerobic pathways | Ensure gas exchange; CO2 supplementation can boost growth in dense algal/protist cultures |
| Food/prey density (for heterotrophs) | Bacterial concentration directly limits growth and observability in bacterivorous protists | Maintain consistent, adequate bacterial food supply when culturing heterotrophic protists |
One thing worth noting from practical culturing work: inoculating a new culture with cells that are already in exponential growth (actively dividing) dramatically shortens the lag phase before growth picks up. Starting with stressed or stationary-phase cells means the new culture wastes time recovering before it can grow. It's the biological equivalent of pitching active sourdough starter versus dried-out flakes.
Why protists can't just keep growing forever

There are hard physical limits on how large a single protist cell can get, and they come down to geometry. As a cell grows larger, its surface area increases as the square of its linear dimension, but its volume increases as the cube. That means the ratio of surface area to volume keeps shrinking. Because nearly everything a cell needs (nutrients, gases, waste removal) has to cross that surface by diffusion, a cell that gets too large simply cannot supply its interior fast enough to sustain itself. This isn't a theoretical concern. It's the same constraint that limits every living cell, from bacteria to your own neurons.
At the population level, growth hits a ceiling for different reasons: nutrient exhaustion, waste accumulation, physical crowding, or predation. In a closed culture flask, protist populations typically follow a recognizable S-curve: a lag phase while cells adjust, an exponential phase where division is rapid, and a stationary phase where growth rate equals death rate and the population stops expanding. Interestingly, the shape of this curve can shift without changing the eventual maximum density. Experiments with Pfiesteria piscicida showed that changing culture container geometry altered the lag phase length while leaving the stationary-phase cell density essentially unchanged. The ceiling was set by something else (resource availability), not by the container shape.
There's also an indirect feedback loop worth knowing about. Heterotrophic protists that feed on bacteria regenerate nitrogen and other nutrients as metabolic byproducts. That nutrient regeneration can actually support additional bacterial growth, which in turn supports more protist growth. So in a mixed microbial system, protist growth is partly self-sustaining through nutrient cycling, right up until the system hits a hard external limit.
This is why protist growth is not fundamentally different from growth constraints in other organisms covered on this site. The same surface-area-to-volume problem, the same nutrient-quota dynamics, and the same population-level ceilings show up whether you're looking at protists, plankton, or larger eukaryotes. Size and resource access impose universal rules.
How to observe, predict, and test protist growth in practice
If you're working with protist cultures, whether for a lab class or genuine research, here are the practical tools and approaches that translate the biology above into something you can actually use.
Counting cells and estimating biomass

The most direct way to track growth is to count cells over time. Hemocytometers are the classic tool for this. For dilute samples (below a few thousand cells per mL), a Nageotte counting chamber works better because it holds a larger volume and gives you statistically reliable counts from sparser samples. For higher throughput, flow cytometry with a stain like LysoTracker Green lets you enumerate heterotrophic protists quickly across abundances from roughly 1,000 to 1,000,000 cells per mL, with counts that compare well to light microscopy.
For photosynthetic protists, optical density (turbidity) and fluorescence are useful proxies for biomass, but both need calibration. Optical density readings depend on cell size, pigment composition, and growth conditions, so you can't assume a fixed conversion factor between OD and dry cell weight across different species or conditions. Fluorescence works similarly: it tracks chlorophyll-related pigments well but varies by species, depth, and even time of day. Nephelometry (measuring scattered light rather than absorbed light) is a simpler, inexpensive alternative that works well for biomass estimation, including in pigmented or opaque samples where standard spectrophotometry struggles.
Predicting growth from conditions
Once you understand the Droop quota framework, you have a mental model for predicting what will happen when you change nutrient supply. Reduce phosphorus availability, and growth rate will drop before cells show any visible stress. Increase it, and growth rate climbs until something else (light, temperature, CO2) becomes the new bottleneck. The same logic applies to any limiting factor: growth rate is controlled by whichever resource is most scarce relative to demand.
For photosynthetic species, watch your pH. In a dense culture with active photosynthesis, pH can drift upward as CO2 is consumed. Once pH climbs above roughly 8.8, growth rate and photosynthesis both start to drop, not because the pH itself is toxic, but because CO2 availability collapses at high pH. This is a common but easily missed cause of growth slowdown in lab cultures.
Recognizing developmental transitions, not just death
If your culture suddenly loses active cells, don't assume they all died. Check for cysts. When conditions change, viruses can switch between replicating and dormant states as part of their life cycle life cycle stages. Encystment can be triggered by temperature shifts, nutrient crash, or even mechanical disturbance, and it can look like a population crash if you're only counting motile cells. Under a microscope, cysts typically appear rounder, less motile, and more refractile than active cells. If you see that pattern, the culture may be recoverable by restoring favorable conditions and allowing excystation to proceed.
- Count cells at regular intervals using a hemocytometer or flow cytometry to build a growth curve and identify which phase the culture is in
- Track pH and temperature daily in photosynthetic cultures, especially as cell density increases
- Inoculate new cultures with exponentially growing cells to minimize lag phase
- Use nutrient-limited experiments (e.g., removing phosphorus or nitrogen) to identify which resource is actually limiting growth rate
- Look for cyst formation when cultures crash unexpectedly, before concluding cells have died
- Calibrate any optical density or fluorescence readings against direct cell counts or dry weight measurements before using them as growth proxies
- Match light cycle duration and intensity to species-specific requirements, especially for circadian-regulated species like dinoflagellates
Protists sit at an interesting crossroads in the biology of growth. They're eukaryotes using recognizably eukaryotic machinery (mitosis, organelles, signaling cascades) but operating as single cells governed by the same diffusion limits and resource constraints that shaped the evolution of every cell-based life form. If you understand how protists grow and develop, you have a solid conceptual foundation for understanding growth across much of life. The same principles that explain why a diatom stops dividing when phosphorus runs out also explain why amoebae and paramecia hit population ceilings, and why even your own cells can't divide indefinitely without resource input. These same nutrient-driven growth and division rules also explain how amoebae grow in size before they split.
FAQ
Is “development” in protists ever like animal development (embryos, organs), or is it always just life-cycle switching?
In most protists it is stage-based change rather than organ building, but you can still see differentiation of cellular form and function between stages. For example, a dividing cell often changes morphology and behavior before cytokinesis, and encystment produces a dormant architecture that is functionally different from the active state.
How can I tell whether growth slowed because of nutrient limitation versus something like light or temperature?
Look for which change shifts the timeline of division. Nutrient limitation usually reduces biomass accumulation and division speed before you see dramatic “death,” while light, temperature, or CO2 bottlenecks often show stronger effects only in photosynthetic species (and sometimes show a day-night timing shift). A practical check is to run parallel cultures that vary one factor at a time while keeping the others matched.
For heterotrophic protist cultures, what role does prey availability play in “growth and development”?
Prey is the nutrient input that drives biomass buildup. If bacterial prey is reduced, protists can enter a stress response that may include stalling growth and, in some groups, switching into cysts. In mixed systems, protists can also regenerate nutrients (like nitrogen) from prey breakdown, so reducing prey can suppress protist growth indirectly by collapsing the nutrient cycling loop.
What does the Droop quota model help me predict in a real culture, and what does it not predict?
It helps you anticipate that growth rate depends on internal stores and that growth can arrest even when external nutrients are not immediately depleted. It does not replace time-resolved observations of the limiting factor, so in practice you still need to track whether division timing and biomass proxies respond to nutrient changes versus light, temperature, or pH.
Why might my protist culture look healthy under the microscope but still stop increasing in number?
The cells can be internally nutrient-starved, meaning biomass accumulation and cell-cycle progression slow even if individual cells still look intact. In other words, “cell-looking fine” does not guarantee “ready to divide,” so tracking division indicators or using time-course counts is more informative than snapshots.
How do I distinguish encystment from cell death during a sudden population crash?
Check for non-motile, rounder, often more refractile cells consistent with cysts. If cyst formation is the cause, restoring favorable conditions should allow excystation and regrowth after a delay. If it is death, cell debris usually increases and viable counts do not recover.
Can physical disturbance or handling steps trigger developmental transitions like encystment?
Yes. Some protists can encyst in response to stressors that include temperature shifts, nutrient drops, or mechanical disturbance. That means centrifugation, major media changes, or rough mixing can sometimes cause a “hidden” population shift into dormant forms even if the culture medium later looks correct.
If I inoculate with cells already in active division, why does the lag phase get shorter?
Because the starting population is already “preloaded” in the cell cycle, with enough biomass and internal nutrient quota to progress toward division. When you start with stressed or stationary cells, they must first rebuild internal stores and complete cell-cycle steps, which creates a longer adjustment period before exponential growth.
What’s a common mistake when using optical density or fluorescence to estimate protist biomass?
Assuming a constant OD-to-biomass conversion across species, growth states, or pigment contents. Cell size, pigment concentration, and even timing of day can change how strongly light absorbance or chlorophyll fluorescence reports biomass, so calibration to your specific species and conditions is crucial.
How can I estimate whether my culture is reaching its population ceiling, not just “slowing down”?
Use a time series to look for an S-curve pattern: early adjustment (lag), rapid increase (exponential), then leveling (stationary where net growth is near zero). A key nuance is that the final maximum density can stay similar even if the lag phase changes, depending on what resource or condition is truly limiting.
Does circadian timing matter for protist division in ways I can observe without lab-grade equipment?
For some photosynthetic protists, division can be gated by the light-dark cycle, so cell counts may rise at predictable times relative to lights on and lights off. Even with basic culturing, keeping the same photoperiod and sampling at consistent times can reveal that division is synchronized to the daily cycle.
Are all protist divisions “classic” mitosis, or should I expect unusual variants?
Expect variation. Many protists use eukaryotic division machinery, but the details can differ substantially, such as unusual chromosome-spindle interactions or differences in whether the nuclear envelope breaks down. If you are interpreting microscopy images, do not assume every protist will show the same visible mitotic sequence.
How Do Paramecium Grow: Growth Cycle, Conditions, and Tips
Learn how paramecium grow: cell cycle growth, binary fission, needed food and water conditions, plus troubleshooting tip