Diatoms grow by photosynthesis and cell division, splitting one cell into two and repeating that cycle as long as they have light, nutrients, dissolved silica, and the right temperature. To actually grow them yourself, you need a starter culture, a nutrient-rich medium (f/2 is the standard), a silicate source if you're culturing marine or silica-hungry species, a light source delivering 50 to 150 µmol photons per square meter per second on a 12:12 light/dark cycle, and water held between 15 and 22°C. Get those conditions right and you'll see visible growth within a week.
How Do Diatoms Grow? Step-by-Step Growth Setup Guide
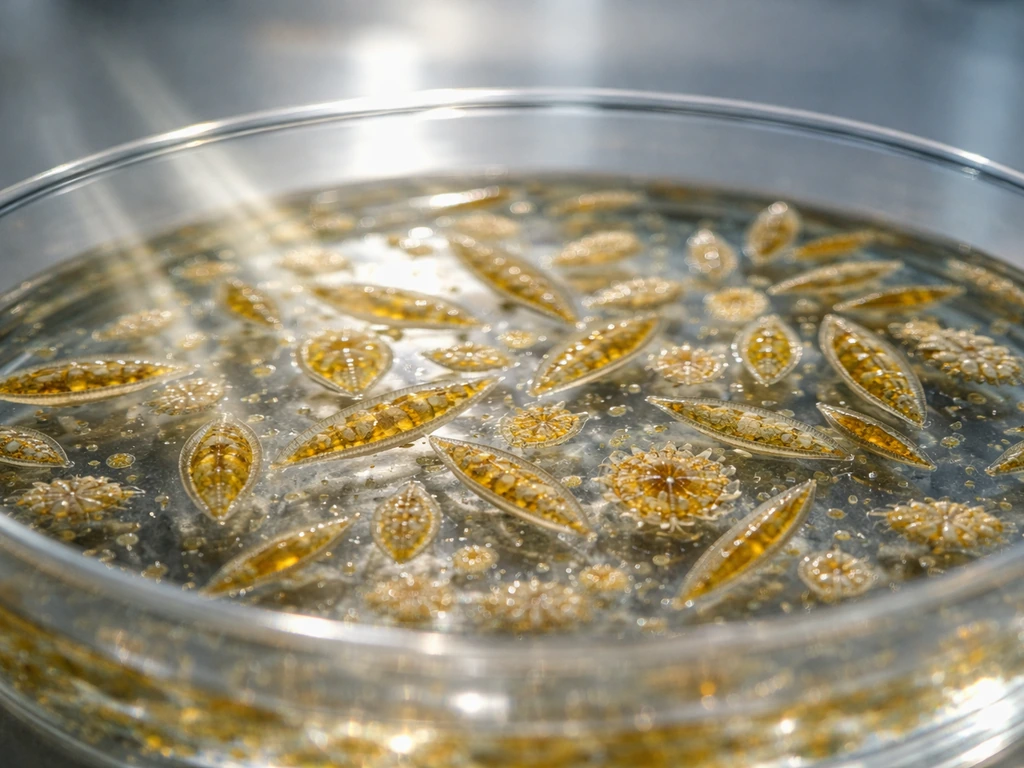
What diatoms actually need to grow (and what stops them)

Diatoms are single-celled algae, and like all algae they grow through photosynthesis and cell division. Each parent cell splits into two daughter cells. Here's the quirk that makes diatoms different from almost every other organism you'll study: each cell is encased in a glass-like silica shell called a frustule, made of two overlapping halves like a petri dish. When a cell divides, each daughter gets one half of the old shell and has to build a new second half. The new half is always slightly smaller. That means with every generation, the average cell size in your culture drifts downward. Over many cycles, cells restore their size through a special sexual or asexual stage called auxospore formation, which effectively resets the size clock.
This biology has a direct practical consequence: dissolved silica is not optional. Silicon limitation can halt the cell cycle at two specific checkpoints, the G1-S boundary (before DNA replication) and the G2-M transition (before the cell actually divides). If there's no silica in the water, diatoms can't build new frustule halves, and division stops cold. Think of silica as the bricks your cells need before they can build the next room. Run out of bricks and construction stops, no matter how much light or nitrogen you provide.
Beyond silica, diatom growth is constrained by the same factors that limit any phytoplankton: light intensity and photoperiod, nitrogen (usually as nitrate), phosphorus (as phosphate), temperature, and space (cell density). These aren't independent dials either. Low light slows photosynthesis, which slows division. Low nutrients starve the metabolic machinery. Overcrowding exhausts nutrients fast and leads to culture crashes. Understanding these levers lets you diagnose problems when growth stalls, which it will at some point.
Starting right: getting a culture and setting up your space
You need a starter culture before anything else. Don't try to collect wild diatoms and hope for the best if you're new to this. You won't know what species you have, and you'll almost certainly introduce competing organisms. Instead, get a known strain from a reputable source. UTEX (University of Texas Culture Collection of Algae) and the Canadian Phycological Culture Centre both supply live diatom cultures along with matching medium recipes. For marine species, Phaeodactylum tricornutum and Thalassiosira weissflogii are beginner-friendly because they're well-documented, fast-growing, and robust. For freshwater work, Fraunhofer's fwDiatom Medium is a published freshwater recipe designed specifically for diatom cultures.
For your vessel, a clear glass Erlenmeyer flask (250 mL to 1 L) works well for beginners. It's easy to mix, easy to monitor visually, and you can loosely stopper it with foam bungs or cotton to allow gas exchange while blocking airborne contamination. You don't need a sterile laminar flow hood, but you do need to work cleanly. Rinse all glassware thoroughly with distilled or deionized water, and ideally autoclave or boil your flasks before use. Whatever contaminants you introduce at the start will outcompete your diatoms later. A little care upfront saves a lot of frustration.
Inoculate your culture to roughly 10,000 cells per mL (1×10⁴ cells/mL) as a starting density. This is a common benchmark used in research settings and gives your diatoms enough neighbors to grow without immediately exhausting nutrients.
Preparing your growth medium

Guillard's f/2 medium is the go-to standard for marine diatom culture, and it's been used reliably since the 1960s. The core recipe includes sodium nitrate at 75 mg/L as the primary nitrogen source, sodium phosphate monobasic at 5 mg/L for phosphorus, EDTA disodium salt at 4.36 mg/L to chelate trace metals and keep them bioavailable, plus a trace metal solution and vitamins including biotin and vitamin B12 at microgram-per-liter concentrations. You can buy premixed f/2 concentrate from PhytoTechnology Laboratories and similar suppliers, or you can make it from scratch using published protocols from the Canadian Phycological Culture Centre.
For diatoms specifically, you must add a silicate source on top of standard f/2. The most common choice is sodium metasilicate. A working range of 10 to 30 mg/L is appropriate for most marine diatom species, and this is why diatom medium is often labeled f/2+Si. Without this addition, your culture will stall even if everything else is perfect. Some aquaculture protocols add silica at the higher end of that range to support dense cultures; for a beginner setup, starting at 15 to 20 mg/L is reasonable.
For freshwater diatoms, the nutrient ratios shift. Use a freshwater-specific recipe like the Fraunhofer fwDiatom Medium, which is calibrated for the lower ionic strength and different trace element availability of freshwater. Don't just dilute marine f/2 and expect it to work for freshwater species.
| Component | f/2 target amount | Notes |
|---|---|---|
| Sodium nitrate | 75 mg/L | Primary nitrogen source |
| Sodium phosphate monobasic | 5 mg/L | Phosphorus source |
| EDTA disodium salt | 4.36 mg/L | Chelates trace metals |
| Sodium metasilicate (Si) | 10–30 mg/L | Diatom-specific; essential for frustule formation |
| Trace metals solution | Per recipe | Includes Fe, Mn, Zn, Cu, Co, Mo |
| Vitamin mix (B12, biotin, thiamine) | µg/L range | Growth cofactors |
Filter-sterilize your prepared medium through a 0.2 µm membrane filter rather than autoclaving if you can, especially the vitamin solution, since high heat degrades vitamins. Store vitamin substock solutions refrigerated and add them to the medium just before inoculation.
Light, temperature, and timing
Light is what powers the whole operation. Diatoms use photosynthesis to fix carbon, and without adequate photon flux, growth slows dramatically. Research shows that 15 µmol photons per square meter per second significantly limits growth in several species including Cylindrotheca and Phaeodactylum. You want to be well above that floor. A target of 50 to 150 µmol photons per square meter per second (PPFD) is a solid working range for most diatom species. Full-spectrum LED grow lights or cool-white fluorescent tubes work well. Keep the light source 15 to 30 cm from the flask and measure intensity with a PAR meter if you have one.
Photoperiod matters as much as intensity. A 12:12 light/dark cycle is the standard starting point and reflects conditions used in published lab studies at 125 µmol photons per square meter per second. Research tracking growth rate as a function of photoperiod shows that diatoms respond predictably: longer light periods generally increase growth rate up to a point, but constant light (24:0) can actually stress some species over time. A simple plug-in timer set to 12 hours on and 12 hours off is all you need.
Temperature sits between 15 and 22°C for most common lab and aquaculture diatom strains. Published experiments confirm growth at 10, 15, and 20°C, with interactions between temperature and photoperiod affecting how fast cells divide. At 22°C under controlled lab conditions, healthy diatom cultures grow reliably. Below 10°C, growth slows sharply. Above 25°C, many species start to struggle. Room temperature (18 to 22°C) works fine for most beginners, as long as you're not culturing in a hot garage in summer.
Expect your culture to show a lag phase of two to four days after inoculation while cells adapt, then enter exponential growth where you'll see the water turn distinctly brown or golden-brown (that's the diatom pigment mix). In a well-set-up flask under these conditions, you can reach visible turbidity within five to seven days.
Keeping the culture healthy: mixing, CO2, and cleanliness

Diatoms sink. Unlike some other phytoplankton, they don't have flagella and can't swim toward the light. If you leave your flask sitting still, cells pile up at the bottom, light penetration drops, and nutrients near the cells get depleted while the rest of the medium is untouched. Gentle swirling or orbital shaking (60 to 80 rpm) keeps cells in suspension and brings fresh medium into contact with them continuously. Even manual swirling twice a day is better than nothing for small hobby setups.
CO2 is the carbon source for photosynthesis, and in a sealed flask it can become limiting as the culture grows dense. For small cultures, leaving the flask loosely stoppered allows atmospheric CO2 exchange and is usually sufficient. For larger or denser cultures, gently bubbling filtered air (0.2 µm filtered to remove contaminants) through the medium supplies both CO2 and mixing. Research shows that CO2 availability interacts with light intensity and growth rate, so in a high-light, high-density culture you may see signs of carbon limitation (rising pH, slowing growth) even with good nutrients.
Cleanliness is an ongoing commitment, not a one-time setup step. Bacteria naturally associate with diatoms and can be beneficial in small numbers, but unchecked bacterial growth competes for nutrients and can produce compounds that harm diatom cells. If you want near-axenic cultures, multiple washes combined with antibiotic selection (antibiotics should be filter-sterilized through 0.2 µm and stored refrigerated) can reduce bacterial load significantly. For hobby purposes, consistent use of clean glassware and sterile medium, combined with regular subculturing, keeps contamination manageable without going full lab-scale.
Troubleshooting when your culture won't cooperate
If your diatoms aren't growing, work through the checklist below systematically. Parasites are different from diatoms, but the same basic question, how do parasites grow, comes down to how they replicate and obtain nutrients in their host environment. Most failures come down to one of four causes: wrong medium, bad light, contamination, or a silica deficit.
- No growth at all after 7+ days: Check that your medium actually contains silicate (if using marine f/2, confirm you added the +Si component). Verify light is reaching the flask at adequate intensity. Make sure temperature isn't too low (below 10°C) or too high (above 25°C).
- Slow growth: Measure light intensity if you can. Values below 30 µmol photons per square meter per second will visibly limit growth. Also check whether the culture is getting mixed, since sedimented cells effectively get no light.
- Green or murky color instead of golden-brown: This usually means contamination by green algae or cyanobacteria outcompeting your diatoms. Discard and start fresh with a cleaner inoculum and better sterile technique.
- Fast initial growth followed by a sudden crash: Classic nutrient exhaustion, most often silicate depletion. The culture bloomed, silica ran out, division stopped, and cells started dying. Start adding semi-continuous dilutions with fresh f/2+Si medium before the crash happens.
- Cloudy medium with no color: Bacterial bloom, not diatom growth. This happens when your medium or glassware wasn't clean. Discard and restart.
- Cells visible but culture not expanding: Check pH. Very dense cultures fix so much CO2 that pH can rise above 9, inhibiting further growth. Increase aeration or dilute the culture.
Scaling up and keeping a healthy culture long-term
The key to a sustainable diatom culture is semi-continuous maintenance: you regularly remove a portion of the culture and replace it with fresh medium before nutrients run out. This prevents the boom-and-bust cycle that kills batch cultures. A common protocol removes 20 to 30 percent of the culture volume every two to three days and replaces it with fresh f/2+Si medium. This keeps cell density in the productive exponential phase and silicate concentrations from bottoming out.
To scale up, move incrementally. Go from a 250 mL flask to a 1 L flask by adding fresh medium and waiting for the culture to grow into the new volume rather than diluting dramatically all at once. Each time you scale, maintain the same light intensity per unit surface area. A culture that's doubled in depth but receives the same lamp from the same distance will have cells in the middle getting far less light, so you either need stronger lighting or shallower vessels as you scale.
Monitoring healthy growth doesn't require a flow cytometer. Visually, a healthy diatom culture deepens from pale yellow-green to rich golden-brown as density increases. Turbidity (cloudiness) rises steadily during exponential growth. If you have a microscope, check for intact frustules and actively dividing cells. At the research level, tools like PAM fluorometry and dissolved oxygen sensors track biomass and photosynthetic activity in real time, but for practical monitoring, color, turbidity, and a weekly microscope check will tell you most of what you need to know.
Keep a spare backup culture at lower density and slightly cooler temperature. If your main culture crashes, and eventually one will, you'll be glad you have a stock to restart from rather than having to order new cells. Label everything with the date and dilution history, especially if you're tracking frustule size over time, since the progressive size reduction across generations is a real phenomenon worth monitoring in longer-term cultures.
How diatom growth compares to other single-celled organisms
If you've looked into how other microorganisms grow, diatom growth has some features that set it apart. Unlike bacteria or simple protists like amoeba or paramecium, diatoms are tied to silica availability in a way no other common microorganism is. Protists show a wide range of growth and developmental strategies depending on their type, but most still rely on cell division and responding to environmental conditions protists like amoeba or paramecium. If you are wondering how paramecium grow, their growth depends on food availability and proper water conditions rather than silica building blocks. If you are comparing growth patterns across single-celled organisms, amoeba growth is driven less by a silica shell and more by food availability and cell division in a suitable environment. Viruses do not grow and develop the way cells do; they replicate only inside living host cells do viruses grow and develop. Their photosynthetic strategy also makes them more like phytoplankton and other algae than like heterotrophic single-celled life. The silica frustule constraint, the size-reduction ratchet across generations, and the tight coupling between cell division and silicon biology make diatoms uniquely interesting to study if you're thinking broadly about how growth works under physical and chemical constraints.
FAQ
Why do my diatoms start growing but then stall after a few days?
If you see only a brief turbidity increase and then it stops, the most common causes are silicate depletion, nutrient depletion, or light not reaching the cells in the middle of the vessel. Check by measuring or replenishing with fresh f/2+Si using a semi-continuous approach (for example, remove 20 to 30% every 2 to 3 days), and verify your PPFD at the liquid surface, not just at the lamp.
Can I grow freshwater diatoms in diluted marine f/2 (with added silica)?
Diatoms grown in seawater-like conditions usually need a diatom medium tailored to that chemistry. Do not swap saltwater diatom medium into freshwater tanks, and do not simply dilute marine f/2. Use a freshwater-specific recipe (like fwDiatom-type media) because ionic strength and trace element availability differ, which can change division rate even when nitrogen, phosphorus, and silica are present.
How do I know if CO2 is limiting my diatom culture?
Yes, leaving CO2 exchange limited can slow growth in denser cultures. A practical rule is to keep the culture loosely stoppered and relatively small at first. If you later increase density or volume, consider gentle bubbling of 0.2 µm filtered air for both carbon supply and mixing, and watch for rising pH plus slowing turbidity as early carbon-limitation signs.
Should I autoclave all my diatom medium and supplements?
Autoclaving is risky for components that include vitamins and other heat-sensitive chemistry. A common workaround is to sterilize the bulk salts by filtration (0.2 µm) and add the vitamin substock after the medium cools. If you have to heat-sterilize, do it only for the non-vitamin portions and keep storage refrigerated, minimizing time before you inoculate.
Why does average cell size seem to keep shrinking in my culture?
Diatoms can form different size distributions depending on whether you are at early exponential growth or later stages. If you want more consistent measurements of frustule size over time, keep a log of your inoculation density and any volume changes, because progressive size reduction is driven by repeated division. Using a semi-continuous culture with regular replacement tends to stabilize growth conditions.
What are the fastest things to check when my diatoms look like they are not growing?
If your culture stays pale and never turns golden-brown, start by confirming silica and light first. Silica can halt division at specific checkpoints, even if nitrate, phosphate, and trace metals are fine. Then check PPFD and mixing, because diatoms sink and the bottom can become light-limited quickly, causing an apparent “no growth” look.
How much shaking is enough, and can too much mixing hurt diatoms?
In small flasks you often want enough mixing to prevent settling without overheating. Use gentle swirling or orbital shaking around 60 to 80 rpm, and avoid splashing onto the neck where contamination risk increases. Also keep light distance consistent, because changes in how you position the lamp can alter PPFD more than you expect.
Do I need antibiotics to keep diatom cultures healthy?
For contamination control, avoid overreliance on antibiotics as a long-term solution. If you attempt near-axenic cultures, reduce bacterial load with clean glassware, sterile medium handling, and multiple washes plus antibiotic selection where appropriate, with antibiotics filter-sterilized and added carefully. For routine hobby culture, consistent sterile technique and regular subculturing usually keep issues manageable.
How should I choose an inoculation density if I do not have a cell counter?
If you are not measuring cells, you can approximate starting conditions by inoculation volume and dilution history, then use color and turbidity trends to judge whether you hit the productive zone. A common target is about 1×10⁴ cells per mL to start, but in practice aim for a visible lag phase (2 to 4 days) followed by steady darkening over about a week, not immediate heavy clouding.
What changes when I scale from a 250 mL flask to 1 L?
Scaling up mainly fails when light intensity per unit area effectively drops or when nutrient and silicate concentrations become imbalanced during early density changes. Move to larger vessels gradually and keep lamp setup geometry the same, or adjust lamp distance and intensity so the liquid receives similar PPFD. Also consider shallower depths or stronger lighting when volumes increase, since cells in deeper regions get far less light.
How Do Protists Grow and Develop: Cell Growth and Division
How protists grow and develop: cell growth, mitosis and division, and life stage changes under nutrient and size limits.


