As you grow from a baby into an adult, your cells mostly increase in number rather than getting bigger. Cell division, specifically mitosis, is the primary engine of body growth. Most of your cells stay within a surprisingly narrow size range throughout your life, held there by physical and biochemical limits. That said, some cell types do get noticeably larger under specific conditions, and understanding which ones, and why, is where things get really interesting.
Do Cells Get Bigger as You Grow? Cell Size vs Number
Cell size vs cell number: the core distinction

When biologists talk about growth, they split it into two fundamentally different things: adding mass to an individual cell (cell growth) and producing more cells through division (cell proliferation). These are not the same process, and they don't always happen together. A tissue can double in size entirely by doubling its cell count while every individual cell stays exactly the same size. That's actually the dominant strategy in most tissues during normal body growth. According to Nature Reviews Molecular Cell Biology, when cell size stays constant, tissue size is controlled almost entirely by regulating how many times cells divide and for how long. So the short version: you're mostly building more copies, not bigger copies.
What's actually happening during early development and childhood
Early development is one of the most counterintuitive phases of growth. Right after fertilization, the embryo goes through rapid rounds of cell division called cleavage, and here's the twist: the total size of the embryo barely changes. Each division cuts cells in half rather than growing new material. In organisms like C. elegans, these cleavage-type divisions produce exponentially more cells that are each progressively smaller than their predecessors. The same pattern shows up in human embryos. Cell number shoots up, cell size actually drops. This is why embryologists sometimes describe early development as a process of parceling out a fixed amount of cytoplasm into more and more packages.
Once you move into fetal development, childhood, and puberty, the pattern shifts: cells do grow before they divide, reaching a threshold size and then splitting. But the net result across tissues is still primarily more cells, not dramatically bigger ones. Organ size during childhood is largely a story of proliferation, with stem and progenitor cells dividing repeatedly to build up tissues and maintain them. In the epidermis, stem and progenitor cells are the ones that grow and divide to replace cells as skin turns over. The cell cycle itself, that cycle of growth, DNA replication, and division, is tuned to keep average cell size relatively stable even as tissues expand rapidly.
When cells do get bigger: hypertrophy and tissue-specific growth

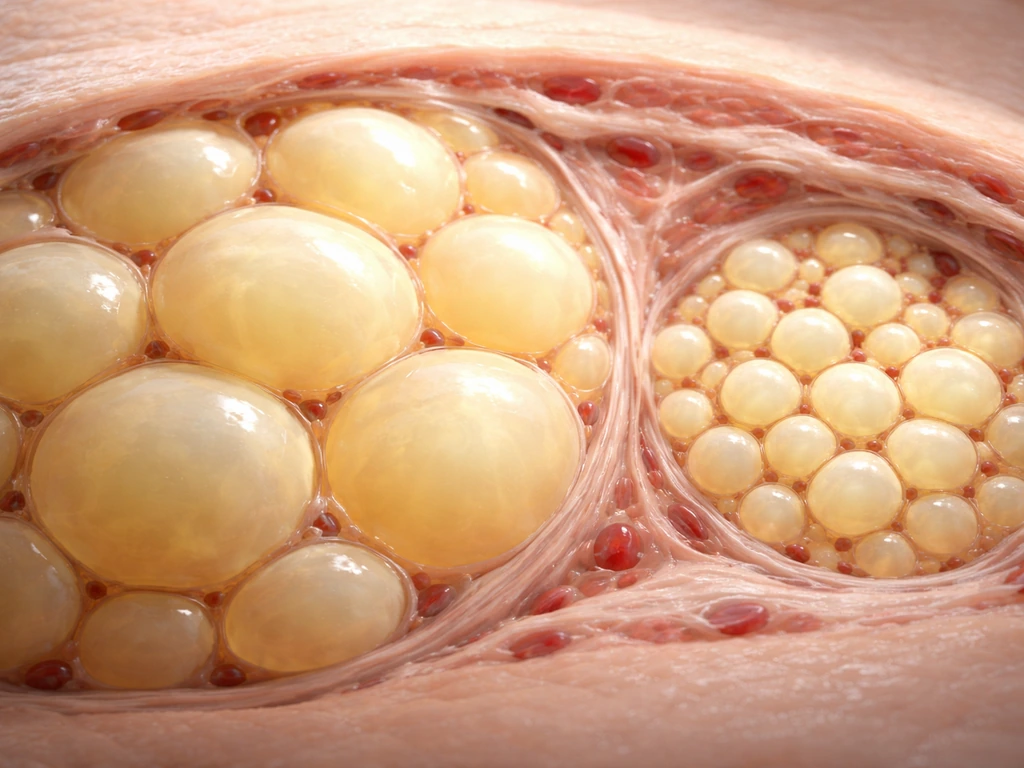
Cell enlargement, called hypertrophy, is real and important in specific contexts. The key is that it's tissue-specific and often condition-specific. It's not the default mode of body growth, but it's a major mechanism in certain tissues, especially ones that don't divide easily or at all in adulthood. Muscle fibers are the classic example: they respond to mechanical overload by getting thicker and longer rather than splitting into more fibers. Fat cells can swell to enormous sizes when storing excess energy. Neurons remodel extensively without dividing. Understanding these exceptions is just as important as understanding the general rule.
Why cells can't just keep getting bigger
There's a fundamental physics problem with making cells too big, and it comes down to surface area versus volume. As any cell gets larger, its volume grows much faster than its surface area. Think of inflating a balloon: the inside space grows much faster than the rubber surface. For a cell, the membrane surface is where nutrients enter and waste exits. If the volume gets too big relative to that surface area, the cell simply can't move molecules in and out fast enough to keep itself alive. Oxygen diffusion is a real bottleneck here, and it's one reason why most cells in your body are microscopic. Biology LibreTexts puts it plainly: as cells increase in size, the surface-area-to-volume ratio decreases, limiting the diffusion the increased volume demands.
Beyond diffusion, the cell's nucleus also has a finite capacity to coordinate gene expression and send instructions to a growing cytoplasm. There's a practical limit to how much cellular territory one nucleus can manage. Cells that do get very large, like muscle fibers, solve this by accumulating multiple nuclei rather than relying on one.
How cell cycle checkpoints keep size in check
Cells don't just grow randomly and hope for the best. The cell cycle has built-in checkpoints that only allow a cell to proceed toward division once it has reached a minimum size and verified that conditions are suitable. Two major molecular pathways govern this: mTORC1 and the Hippo pathway. mTORC1 acts like a growth sensor, promoting protein synthesis and mass accumulation when nutrients and energy are available. The Hippo pathway works as a brake, restricting proliferation and even triggering programmed cell death when organ size gets too large. When Hippo signaling is dysregulated, organs can overgrow, which is one reason this pathway is studied closely in cancer research. If you're wondering why do cells grow in the first place, this cell size homeostasis and checkpoint logic is the starting point.
Research on mammalian cell populations shows that cells maintain size homeostasis by adjusting both how fast they grow and how long they spend in each phase of the cell cycle. Some cells behave as "sizers," dividing only after reaching a target size. Others behave as "adders," adding a fixed amount of mass before dividing regardless of starting size. The result in both cases is that average cell size in a tissue stays relatively stable over time, preventing runaway enlargement.
Real examples by cell type
Muscle fibers: the hypertrophy poster child

Skeletal muscle fibers are genuinely large cells, and they get bigger in response to resistance training. When mechanical overload is applied repeatedly, mTORC1 signaling ramps up protein synthesis, ribosomes multiply, and satellite cells (muscle stem cells) donate additional nuclei to the growing fiber in a process called myonuclear accretion. The fiber swells in cross-sectional area, which is what drives visible muscle growth. This is a case where cell size increase is the primary mechanism, not adding more fibers.
Fat cells: size and number both matter
Adipocytes (fat cells) are fascinating because they can expand via both hypertrophy and hyperplasia, depending on the situation. How does adipose tissue grow? Epithelial tissue growth follows related principles of cell division and size control, with specific pathways determining how quickly epithelial layers expand How does adipose tissue grow?. In obesity, it can expand by fat cells enlarging and, especially during earlier life, by adding more fat cells through hyperplasia Adipocytes (fat cells). In adults, excess caloric intake initially causes fat cells to swell with lipid droplets, sometimes reaching several hundred micrometers in diameter. But adipocyte number is largely set during childhood and adolescence, and that number remains relatively stable in adults. This is why childhood obesity is a particular concern: it can drive hyperplasia (more fat cells formed early), and those cells stick around. Adult weight changes are more about fat cell size than fat cell number, though both mechanisms are possible under extreme conditions.
Neurons: growth without division
Most mature neurons in your brain are postmitotic, meaning they've permanently exited the cell cycle and won't divide again. When neurons do try to re-enter the cell cycle, it often leads to cell death rather than successful division. Instead, neurons grow by extending dendrites and axons, forming new synaptic connections, and remodeling their structure in response to experience. Adult neurogenesis does occur in a few specific brain regions, but it's limited and comes from a small pool of adult neural stem cells. For the vast majority of your neurons, growth means structural remodeling, not adding more neurons or dramatically changing soma size.
Red blood cells: constant turnover, stable size
Red blood cells (RBCs) are a completely different story. They survive about 120 days and are continuously replaced at a rate of roughly 1/120th of your total count every day. They don't get bigger as you age in any dramatic way, though some studies using Coulter counter measurements have found that mean corpuscular volume (MCV, the average RBC size) does increase slightly with age. These shifts are modest and relate more to turnover kinetics and bone marrow dynamics than to any growth-driven enlargement. RBCs are actually a good example of a cell type where size stays tightly controlled and turnover, not growth, is the key maintenance mechanism.
| Cell/Tissue Type | Primary Growth Mechanism | Does Size Increase? | Key Regulator |
|---|---|---|---|
| Skeletal muscle | Hypertrophy (fiber enlargement + myonuclear accretion) | Yes, significantly | mTORC1, satellite cells |
| Fat (adipose) | Hypertrophy in adults; hyperplasia mainly in childhood | Yes, substantially | Caloric balance, developmental timing |
| Neurons | Structural remodeling (dendrites, axons) | Minimal size change | Synaptic activity, growth factors |
| Red blood cells | Continuous turnover and replacement | Slight MCV increase with age | Bone marrow, erythropoietin |
| Skin/epithelium | Rapid proliferation (cell division) | Not significantly | Hippo pathway, growth factors |
| Early embryo cells | Cleavage divisions (cell number up, size down) | Cells get smaller | Maternal-to-zygotic transition |
How to think about this like a growth biologist
If you're studying growth biology, or just trying to reason through a question like this on an exam or in a lab, here's a practical mental checklist. The question "do cells get bigger? In many tissues, cells do not keep growing indefinitely, because growth is balanced against division and tight size limits cells do grow. " almost always needs a follow-up: bigger in which tissue, under which conditions, and at which stage of development?
- Ask whether the tissue in question is still actively dividing. Proliferating tissues (skin, gut lining, bone marrow) grow mainly by adding cells, not by enlarging them.
- Check whether the tissue is postmitotic or terminally differentiated. Neurons and mature muscle fibers can't easily divide, so any growth has to come through structural enlargement or remodeling.
- Consider the stimulus. Exercise drives muscle hypertrophy. Caloric surplus drives adipocyte swelling. Without a specific trigger, most adult cells don't spontaneously enlarge.
- Think about surface-area-to-volume constraints. If a cell type is already large, there's likely a compensating mechanism (multiple nuclei, lots of mitochondria, specialized transport) keeping it functional.
- Look at the developmental stage. In embryos, cleavage means more cells and smaller size. In childhood and puberty, proliferation dominates body growth. In adulthood, tissue maintenance shifts toward turnover and hypertrophy in specific contexts.
- Watch for pathway clues. mTORC1 activity signals growth and mass accumulation. Hippo pathway activation signals size restraint and proliferation limits. Both pathways are targets of active research.
If you're looking at microscopy data or tissue sections and trying to interpret what you see, cell size uniformity in a tissue usually points to proliferation-driven growth. If you see cells with dramatically different sizes, especially very large cells alongside normal-sized ones, that's a clue that hypertrophy or some abnormal growth event is happening. Adipose tissue from an obese individual, for example, shows noticeably larger adipocytes under a microscope compared to lean tissue.
The big misconception to avoid is the intuitive but wrong idea that you grow because your cells grow. That's not the primary story. You grow because you make more cells, with cell size carefully regulated by checkpoints, nutrient sensors, and physical constraints. Cell enlargement is the exception, not the rule, and when it does happen, it's almost always in a tissue where division is limited and a specific environmental trigger is present. Keep that distinction sharp and the rest of growth biology falls into place much more cleanly.
FAQ
So do cells ever get bigger in adulthood, or is it only during early development?
Some do get bigger in adulthood, but usually only in specific tissues and triggers. Classic examples are skeletal muscle fibers after resistance training (hypertrophy with added nuclei) and adipocytes during periods of energy surplus (swelling of existing cells, and sometimes expansion earlier in life via increased cell number). For many other tissues, average cell size stays fairly stable because division rates and cell-cycle checkpoints compensate.
If organ size increases during growth, why are cell sizes often unchanged?
Organ size can rise mainly through adding more cells rather than enlarging each cell. When the average cell size is held near a steady target, changing tissue dimensions largely comes from increasing cell number (cell proliferation duration and division rate) rather than bulk cell enlargement.
What does it mean when people say cells have “size homeostasis” in a tissue?
It means cells in a tissue tend to correct for being too small or too large by adjusting cell-cycle timing and growth rate. In practice, cells may behave like “sizers” (dividing only after reaching a target size) or “adders” (adding a fixed increment of mass before division). Either way, the population’s average size doesn’t drift upward unchecked.
Why can’t cells just keep getting larger to support growth?
Two constraints kick in quickly. Surface area limits nutrient and waste exchange (diffusion slows when volume grows faster than membrane area), and the nucleus can only coordinate gene expression for a limited amount of cytoplasm. Large cells often solve this by using multiple nuclei, as muscle fibers do with myonuclear accretion.
How can I tell hypertrophy versus proliferation when looking at tissue under a microscope?
A useful clue is the distribution of cell sizes. If average cell size shifts upward (or you see consistently larger cells), hypertrophy is likely. If cell sizes are fairly uniform but tissue mass or cell counts rise, proliferation is likely. Mixed patterns can occur, so examining multiple markers and time points helps.
Do cancer cells follow the same rule about cell size and division?
Cancer can disrupt normal size control and checkpoint logic, which can lead to abnormal growth and altered cell size distributions. Pathway dysregulation, especially in signaling systems that restrain organ and tissue size, can decouple proliferation from proper size-checkpoint requirements, so tumor cells may not show the usual tight size stability.
Why do embryos show decreasing cell size with rapid divisions?
Early development features cleavage-like divisions where cells split without substantial net growth of total embryo mass. Each division partitions existing cytoplasm into more packages, so cells get smaller even as total cell number rises rapidly.
Do red blood cells get bigger as you get older?
Not dramatically. Mean RBC size can shift slightly with age in some measurements, but most of what changes with aging relates to turnover and marrow dynamics rather than a growth program that makes RBCs substantially larger. The dominant pattern is continual replacement rather than enlargement.
Are neurons supposed to enlarge as you mature?
Most mature neurons are postmitotic, so they do not grow by dividing. Instead, they remodel structure by extending dendrites and axons and changing synaptic connectivity. “Growth” in neurons usually means wiring and branching changes, not a large sustained increase in soma size.
If muscle fibers get thicker with training, does that mean more muscle cells are added?
Typically not in the way that tissue proliferation does. Resistance training mainly increases fiber size through hypertrophy, including added nuclei from satellite cells to support the larger cytoplasm. Muscle growth is driven primarily by increased cross-sectional area of existing fibers, not creating many new fibers.
Why is childhood obesity often described as more concerning than adult weight gain?
Because early-life conditions can increase adipocyte number through hyperplasia, creating more fat cells that persist. Adult weight gain is more often associated with fat cell enlargement (hypertrophy) than large increases in cell count, so the long-term risk differs depending on when the expansion occurs.
What is the most common mistake when answering “do cells get bigger as you grow?”
Assuming growth of your body automatically means your cells grow. In most tissues, body growth is dominated by making more cells while keeping individual cell size relatively regulated. Cell enlargement is real but mainly occurs under specific conditions, such as in certain nondividing or division-limited contexts.
How Does Adipose Tissue Grow: Mechanisms, Signals, Limits
See how adipose expands via fat-cell enlargement or new-cell formation, driven by insulin and energy surplus, limited by