Adipose tissue grows in two ways: fat cells get bigger, or new fat cells form. That is the short answer. The longer answer is that which of those two paths dominates depends on how much energy surplus you are running, how long it has been going on, where in the body the tissue lives, and what your hormones, genes, and even your local oxygen supply are doing at any given moment. Let's walk through all of it, because understanding the mechanism is genuinely useful for understanding how growth works in living systems.
How Does Adipose Tissue Grow: Mechanisms, Signals, Limits
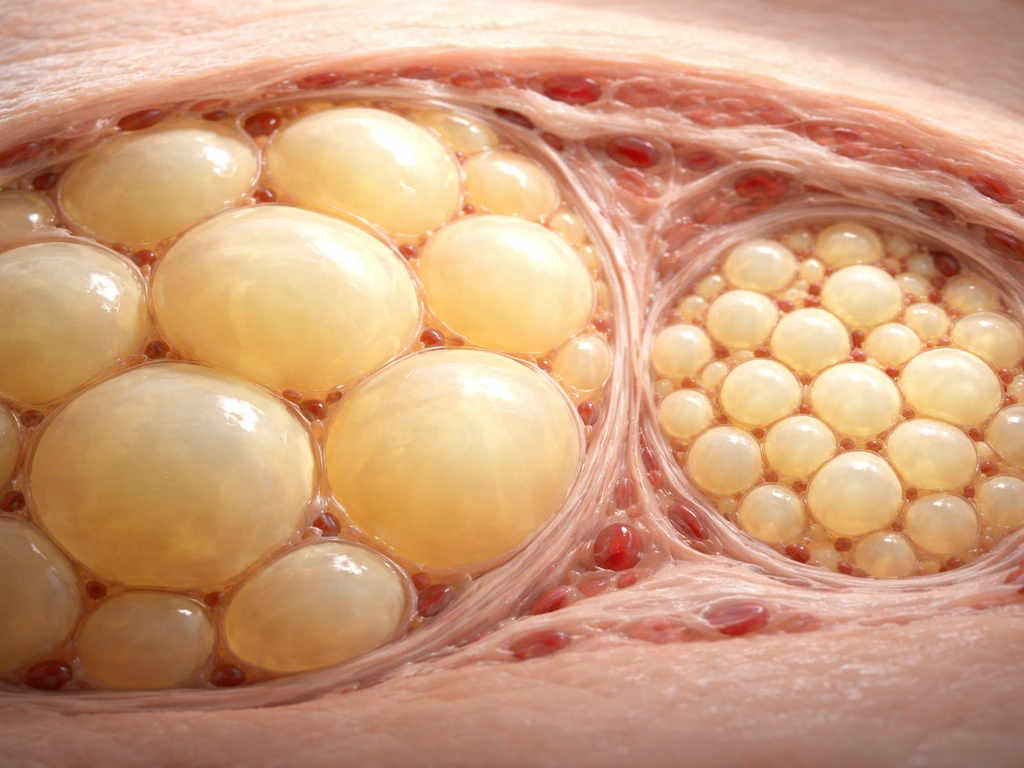
Two ways adipose tissue actually expands: hypertrophy vs. hyperplasia

Think of a storage warehouse. When you need more space, you have two options: pack each existing room more tightly, or build new rooms. Adipose tissue does exactly the same thing. Hypertrophy means individual fat cells (adipocytes) swell larger as they fill with lipid droplets. Hyperplasia means new fat cells are generated from precursor cells, increasing the total number of adipocytes in the tissue.
Research points to a rough sequence: early in fat mass gain, hypertrophy dominates. Existing adipocytes simply pack in more triglyceride. But cells cannot expand indefinitely. A multi-stage model of obesity progression proposes that as hypertrophy reaches its limits, some of the largest cells undergo a kind of necrotic wave, essentially failing under the pressure of overexpansion, and that this stress signal partially triggers the recruitment of new adipocytes through hyperplasia. In other words, the tissue uses enlargement first because it is faster, then recruits new cells when enlargement alone cannot keep up with the storage demand.
Why does this matter? Because the two pathways are not metabolically equivalent. Evidence from lineage fate-mapping studies in animals suggests that a tissue expanding mostly through hyperplasia (more, smaller cells) tends to be metabolically healthier than one expanding purely through hypertrophy (fewer, enormously enlarged cells). Large, hypertrophied adipocytes become stressed, hypoxic, and inflammatory, which is when the biological trouble starts. That said, the relationship is context-dependent: some research shows that people whose fat tissue is predominantly hypertrophic show larger improvements in insulin sensitivity after weight loss, likely because there is more room for the cells to shrink.
How fat cells are actually born: the process of adipogenesis
New adipocytes do not appear from nowhere. They develop from precursor cells called preadipocytes, which live within the stromal-vascular fraction of adipose tissue. The transition from preadipocyte to mature, lipid-loaded adipocyte is called adipogenesis, and it is a tightly controlled gene expression program with several checkpoints.
The transcription factor cascade that kicks it all off

The two master regulators of adipogenesis are PPARγ (peroxisome proliferator-activated receptor gamma) and C/EBPα (CCAAT/enhancer-binding protein alpha). Together they switch on the full fat-cell gene program. But before those two get going, an earlier player called C/EBPβ has to set the stage. C/EBPβ acts early in the cascade, helping to drive up C/EBPα and PPARγ2 expression. If you block C/EBPβ, the downstream program stalls and adipogenesis fails to launch.
Sitting upstream of all of this is the Wnt/β-catenin signaling pathway, which acts as a brake on differentiation. When Wnt signaling is active, β-catenin accumulates in the nucleus and suppresses the adipogenic program by binding TCF/LEF transcription factors. For a preadipocyte to commit to becoming a fat cell, nuclear β-catenin has to come down. This is one reason inflammatory signals like TNF-α, which can sustain β-catenin signaling, actually inhibit adipogenesis: they keep the brake on.
Glucocorticoids play a permissive role here. They promote C/EBPβ acetylation and help relieve repressive chromatin complexes, nudging preadipocytes toward commitment. They also drive expression of KLF15, another transcriptional supporter of the adipogenic program. So stress hormones, chronically elevated, can paradoxically push the body toward greater fat cell formation, a detail worth holding onto.
The role of cell division in early differentiation
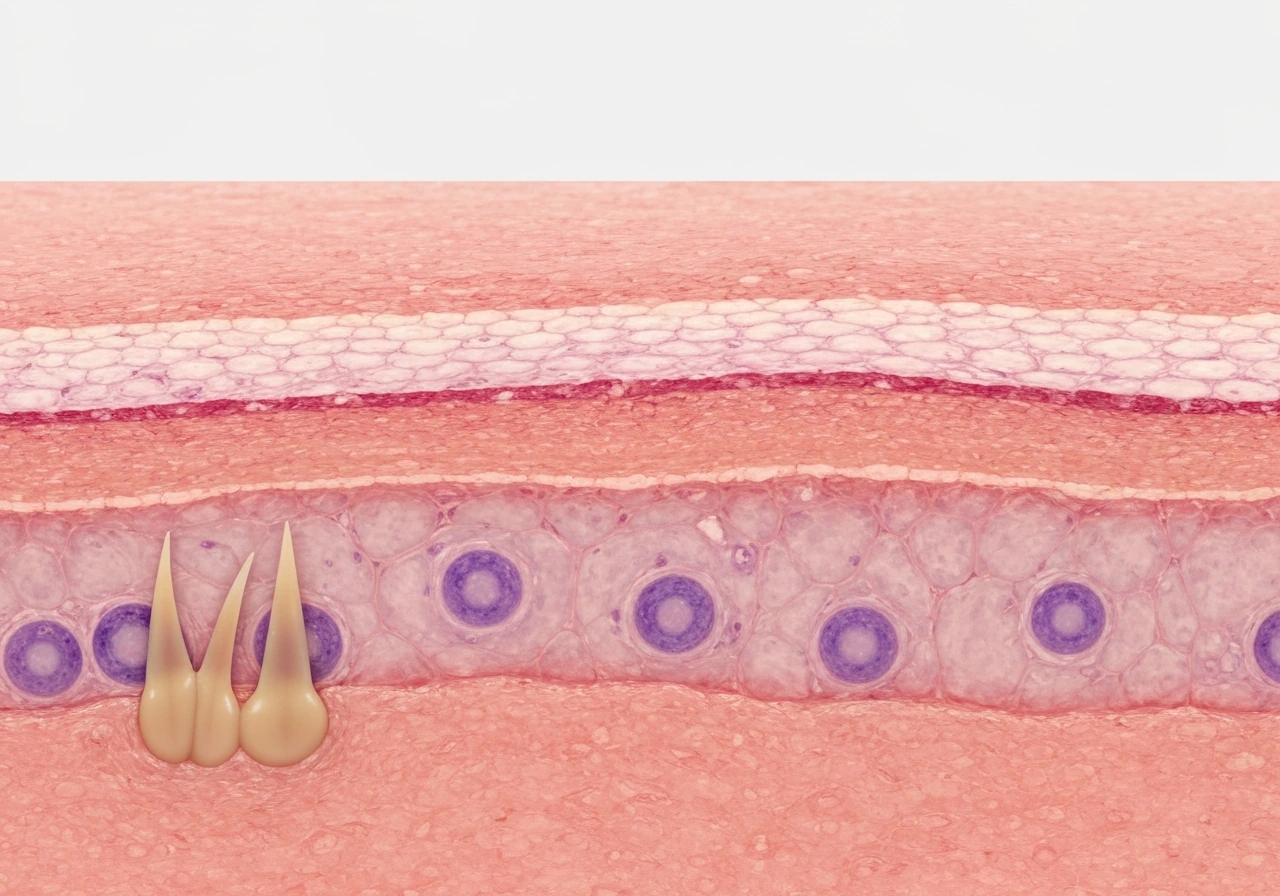
Before a preadipocyte fully differentiates, it goes through a round of cell division called mitotic clonal expansion. In the epidermis, a similar balance of cell division controls which cells grow and divide as the tissue renews cell division called mitotic clonal expansion. This brief proliferative burst appears to be necessary for the differentiation program to complete. The RNA demethylase FTO influences this process: higher FTO activity accelerates mitotic clonal expansion and increases adipocyte formation, while blocking FTO stalls it. This is one reason FTO genetic variants are associated with obesity risk in humans. The connection to cell division is a nice reminder that adipogenesis is not just a metabolic story but also a cell biology story, closely related to how cells grow and divide more generally.
Hormones and metabolic signals: what tells fat tissue to grow
Adipose tissue does not expand on its own initiative. Understanding how a cell grows also depends on the same balance of signals and constraints that govern adipose expansion how does a cell grow. It responds to signals, most of which converge on a few key hormones.
Insulin: the primary driver
Insulin is the dominant pro-storage hormone. When blood glucose rises after a meal, the pancreas releases insulin, which drives glucose into cells, promotes fatty acid uptake into adipocytes, and actively suppresses the breakdown of stored fat (lipolysis). In adipose tissue specifically, insulin stimulates the enzyme lipoprotein lipase, which pulls triglycerides out of circulating lipoproteins and shuttles fatty acids into fat cells for storage. The more frequently and severely insulin is elevated, the more time adipose tissue spends in storage mode rather than breakdown mode.
Glucocorticoids: a complicated supporting role
Cortisol and other glucocorticoids are lipogenic in adipose tissue and also recruit adipose progenitors, as discussed above. Chronically elevated cortisol (from stress, poor sleep, or conditions like Cushing's syndrome) can promote fat gain, particularly in visceral depots. However, glucocorticoids also impair glucose transport and lipid homeostasis in mature adipocytes, creating a somewhat paradoxical picture. They accelerate differentiation of new fat cells while simultaneously making existing ones less metabolically efficient.
Catecholamines: the counter-signal
Adrenaline (epinephrine) and noradrenaline do the opposite of insulin. They bind β-adrenergic receptors on adipocytes, raise intracellular cyclic AMP, and activate hormone-sensitive lipase (HSL) through phosphorylation, triggering lipolysis: the release of stored fatty acids back into circulation. ATGL (adipose triglyceride lipase) handles a large portion of basal lipolysis as well. When catecholamines are high and insulin is low, fat tissue is in breakdown mode, not growth mode. This is the state you see during fasting, exercise, or acute stress.
Nutrients, energy balance, and how diet actually fuels adipose growth
Fat tissue expands when energy intake consistently exceeds energy expenditure. That is the thermodynamic reality. But the route by which dietary nutrients get converted into stored fat is worth understanding in more detail.
Carbohydrates drive insulin release more potently than fats or proteins. When carbohydrate-rich meals push blood glucose high repeatedly, insulin stays elevated for longer stretches of the day. This keeps adipose tissue in storage mode and suppresses lipolysis. Dietary fats, once digested into fatty acids and packaged into chylomicrons, are also taken up by adipose tissue via lipoprotein lipase, which is itself upregulated by insulin. So carbohydrates and dietary fats work together: carbs spike the insulin that enables fat storage, and dietary fat provides the substrate being stored.
Under sustained energy surplus, adipocytes become hypertrophic while the tissue simultaneously pursues hyperplasia to expand storage capacity. This is actually a protective mechanism: adipose tissue is trying to buffer excess circulating glucose and fatty acids, keeping them out of the bloodstream where they would be toxic to other organs. Visceral and ectopic fat depots (around the liver, heart, and muscles) accumulate when the subcutaneous adipose tissue's buffering capacity is exhausted.
What stops fat tissue from expanding forever
This is where the biology gets genuinely interesting, because it connects adipose growth to the broader question of what limits growth in any living system. If you want the big-picture mechanism behind why do cells grow, it helps to see adipose growth as a related example of how cells expand under signals and constraints. These same constraints and signaling pathways are part of how cells grow or increase in size, not just how fat tissue expands. Cells cannot simply expand without bound, and neither can tissues.
Cell size limits and structural remodeling
As adipocytes enlarge, they undergo dramatic internal reorganization. Research shows that adipocyte expansion involves drastic actin remodeling and increased Rho-kinase activity, essentially restructuring the cell's cytoskeleton to accommodate the growing lipid droplet. This is metabolically costly and physically stressful. At some point, cells hit a physical ceiling. Extremely enlarged adipocytes are fragile, prone to rupture, and they begin to die. The necrotic wave mentioned earlier in obesity progression is a direct consequence of cells being pushed past their structural limits.
Extracellular matrix and fibrosis
Adipose tissue is not just cells. It is embedded in an extracellular matrix (ECM) of collagen and other structural proteins. For the tissue to expand healthily, the ECM has to remodel alongside it: breaking down old matrix and rebuilding it in a more permissive configuration. When this remodeling fails, as it does during rapid or excessive expansion, the matrix becomes stiff and fibrotic, physically caging the adipocytes and preventing further healthy expansion. Excessive ECM deposition also impairs the angiogenic response, meaning blood vessel growth cannot keep pace.
Oxygen supply and the hypoxia problem
Blood vessels need to grow alongside expanding tissue to maintain oxygen delivery. In rapidly expanding adipose tissue, vascular growth often lags behind. The result is local hypoxia: pockets of fat tissue with inadequate oxygen supply. Oxygen-deprived adipocytes activate a transcription factor called HIF-1α (hypoxia-inducible factor 1-alpha), which triggers inflammatory programs and fibrosis pathways. Early in high-fat diet feeding, uncoupled adipocyte respiration actually increases oxygen consumption, making the tissue relatively hypoxic even before gross obesity sets in. This HIF-1α activation is strongly linked to adipose inflammation and the development of insulin resistance. So the physical constraint of oxygen supply is not just a passive limit on growth; it is an active driver of tissue dysfunction.
Inflammation as a growth disruptor
As hypertrophied adipocytes become stressed and begin to die, they attract macrophages. These immune cells surround dead or dying fat cells in structures called crown-like structures (CLS). The macrophages release inflammatory cytokines including TNF-α and IL-1β. TNF-α, as noted earlier, directly inhibits adipogenesis by sustaining the Wnt/β-catenin brake on preadipocyte differentiation. This creates a feedback loop: overstretched tissue triggers inflammation, inflammation blocks the formation of new, healthy adipocytes, and the existing adipocytes become even more metabolically impaired. It is one of the clearest examples of how a growth-limiting signal in a tissue can cascade into broader systemic dysfunction.
Why some people store more fat than others
Given the same diet and activity level, different people accumulate fat at different rates and in different places. The reasons touch on genetics, age, sex, and environment, and they are not trivial.
| Factor | How it influences adipose growth | Practical implication |
|---|---|---|
| Genetics (BMI loci) | Variants like FTO regulate adipogenesis through cell-cycle control; other loci affect appetite and energy expenditure | Baseline predisposition to fat storage capacity is partly heritable |
| Genetics (fat distribution) | 13+ genetic loci independently shape waist-hip ratio with distinct architecture from BMI loci; strong sexual dimorphism in these variants | Where fat accumulates is partly genetically programmed, not just a function of total fat mass |
| Sex | Estrogen promotes subcutaneous storage; androgens favor visceral deposition; fat distribution genetics are sexually dimorphic | Women tend toward subcutaneous (hip/thigh) fat; men toward visceral (abdominal) fat |
| Age | Preadipocyte differentiation capacity declines with age; adipose remodeling becomes less efficient; glucocorticoid sensitivity changes | Older adults may accumulate more dysfunctional fat even at lower total mass |
| Chronic stress / cortisol | Glucocorticoids promote fat cell recruitment and lipogenic activity, especially visceral | Prolonged stress or poor sleep can drive visceral fat gain independent of diet |
| Physical activity | Exercise promotes catecholamine-driven lipolysis and can induce beige adipocyte formation in subcutaneous depots via PRDM16 pathways | Regular movement shifts adipose biology toward a more thermogenic, less inflammatory state |
Subcutaneous vs. visceral fat: two very different growth stories
Not all adipose tissue is the same, and the depot in which fat accumulates matters enormously for both metabolic health and the biology of how growth proceeds.
Subcutaneous adipose tissue
Subcutaneous fat, found just under the skin on the hips, thighs, and abdomen, is generally the body's preferred storage depot and the metabolically safer one. It has a higher capacity for hyperplastic expansion (generating new adipocytes) and is more responsive to insulin in a healthy way. Subcutaneous adipose tissue, particularly in the thigh and hip regions, can also harbor beige adipocytes: cells that express the thermogenic protein UCP1 under conditions like cold exposure or exercise. The transcription factor PRDM16 is a key determinant of this thermogenic identity in subcutaneous depots, powerfully activating brown-fat-like programs including PGC-1α and mitochondrial gene expression. This ability to shift toward energy burning rather than pure storage is largely a subcutaneous depot feature.
Subcutaneous adipose expansion has limits, though. Research has documented a saturation point, particularly during and after puberty, where subcutaneous storage capacity becomes functionally full. Once that capacity is exceeded, fat begins to accumulate in visceral and ectopic depots, and metabolic dysfunction rises sharply. This saturation concept is important: it means that the subcutaneous depot acts as a protective buffer, and its failure is a key transition point toward metabolic disease.
Visceral adipose tissue
Visceral fat sits deep in the abdominal cavity, surrounding the organs, and drains directly into the portal vein supplying the liver. It is metabolically more active in terms of lipolysis, which means it releases fatty acids into the circulation more readily. It is also more prone to inflammation: associations between adipose inflammation and insulin resistance are stronger and more consistent for visceral than subcutaneous fat. Visceral fat shows higher macrophage infiltration, more crown-like structures, elevated TNF-α and IL-6 production, and lower levels of adiponectin (an insulin-sensitizing adipokine). The proliferative potential of progenitor cells also appears depot-specific, with certain progenitor compartments in different fat depots showing distinct capacities for hyperplastic expansion.
The practical upshot: abdominal girth and waist-to-hip ratio are more informative about metabolic risk than total body weight alone, precisely because they reflect visceral versus subcutaneous depot balance.
What to actually pay attention to
If you are trying to understand adipose growth in a real-world context, here is what the biology tells you to focus on. First, energy surplus duration matters more than any single meal. Sustained excess is what drives the hypertrophy-to-hyperplasia transition. Second, insulin behavior over time is the key metabolic lever: how frequently insulin is elevated, and how sensitive tissues are to it, shapes how much time adipose tissue spends in storage mode. Third, fat distribution (where on the body adipose tissue is accumulating) is a better signal of biological risk than total fat mass, because visceral expansion carries a distinct inflammatory and metabolic signature. Fourth, lifestyle factors that support subcutaneous rather than visceral growth, and that promote the beige thermogenic program in subcutaneous fat, such as regular physical activity and avoiding chronic stress, genuinely alter the biology at the cellular level.
The biological red flags to watch for are the ones that signal the tissue has shifted from healthy expansion to impaired remodeling: increasing fibrosis, chronic low-grade inflammation (flagged by markers like elevated CRP or adipokine imbalance), and the overflow of fat into ectopic sites like the liver and muscle. Those changes indicate the tissue's growth machinery is no longer working well, and that is where understanding the cell-level mechanisms, covered throughout this article, becomes practically meaningful.
Adipose tissue growth is a fascinating example of how biological growth is never just about getting bigger. It is about coordinated signaling, structural limits, and the competition between healthy expansion and tissue dysfunction. The same themes show up across living systems whether you are looking at how epithelial tissue grows, how individual cells increase in size, or why cells cannot simply divide and expand without limit. If you are wondering about cell size changes as growth happens, the question of whether cells get bigger as you grow is a useful one to keep in mind alongside adipose hypertrophy do cells get bigger as you grow. Those growth principles connect to how epithelial tissue grows, too, through tightly regulated signaling and cell-cycle control. It is also a direct question of how cells grow in size and number, which is exactly the kind of control adipose tissue reveals do cells grow. Adipose tissue just makes those themes unusually visible because its growth, and its failure to grow well, has such direct consequences for whole-body health.
FAQ
Does adipose tissue grow the same way in everyone?
No. The balance between hypertrophy and hyperplasia differs by depot (subcutaneous vs visceral), age (capacity often shifts around puberty), and chronic hormone and inflammatory context. Two people can gain the same total fat, yet one may expand mainly by creating new adipocytes, while the other relies more on enlarging existing ones, which changes metabolic risk.
Is it possible to gain fat even if insulin does not stay “high” all day?
Yes. Acute insulin spikes after meals matter, but so do other drivers like glucocorticoids, sleep disruption, and reduced energy expenditure. Also, insulin sensitivity varies by person, so the same insulin level can produce different storage signaling and different lipolysis suppression.
How can I tell whether fat gain is due to hypertrophy or hyperplasia?
You cannot directly measure it at home, but surrogate clues exist. Metabolic dysfunction with high inflammation and increasing ectopic fat (fat in liver or muscle) tends to align more with impaired healthy expansion, which often involves more problematic hypertrophy and failure to recruit new cells effectively.
Why does rapid weight gain or “surplus” seem to worsen inflammation quickly?
When expansion outpaces vascular remodeling, local hypoxia can develop, activating HIF-1α and inflammatory and fibrosis programs. That can happen even before major body-weight changes, especially early with high-fat diet patterns that increase adipocyte oxygen demand and worsen tissue oxygen balance.
What does “subcutaneous fat is safer” mean, and when is it not true?
It means subcutaneous depots typically have greater capacity to expand through hyperplasia and can support a thermogenic, beige shift (UCP1 program) under the right cues. It is not always protective, though, if subcutaneous storage saturates, fibrosis rises, or fat starts spilling into visceral or ectopic sites.
Can stress hormones increase fat cell number, not just fat size?
Yes. Chronic glucocorticoid signaling can promote differentiation of new adipocytes by supporting early adipogenic transcriptional steps, so it can increase adipocyte formation. At the same time, glucocorticoids can reduce metabolic efficiency in mature adipocytes, creating a mixed effect (more cells, but less favorable function).
Why would inflammation inhibit the creation of new adipocytes?
Inflammatory cytokines can sustain the differentiation “brake” pathway you highlighted earlier, especially by keeping Wnt/β-catenin signaling active in precursor cells. That means overstretched, stressed fat cells can both arise from failed expansion and also prevent the tissue from repairing itself by generating new, healthier adipocytes.
Do exercise and cold exposure change adipose growth, or only energy use?
They can change the tissue program. Activity and cold can favor a subcutaneous beige-like identity through regulatory pathways such as PRDM16-dependent thermogenic gene activation, which shifts the depot toward energy burning rather than pure storage, changing how new adipocytes contribute to overall behavior.
What is a common mistake when thinking about fat gain mechanisms?
Assuming one meal or one hormone explains everything. Adipose growth depends on duration of surplus, repeated insulin patterns, and the tissue’s remodeling and oxygen supply capacity over time. Short bursts of intake matter less than the cumulative imbalance and the inflammatory or hormonal environment.
If adipocytes can die when they are too large, why doesn’t the body just stop expanding immediately?
Because continued energy surplus keeps storage pressure high, and the tissue tries to compensate by recruiting new adipocytes. When fibrosis, oxygen limitation, and inflammatory feedback loops prevent healthy remodeling, the “repair” response becomes less effective, so dysfunction can progress instead of stopping.
Should waist-to-hip ratio always be the top metric for adipose growth risk?
It is a useful risk indicator because it reflects visceral versus subcutaneous balance, but context matters. Some people have lower waist metrics yet still show ectopic fat accumulation, for example if insulin resistance is present for other reasons. If possible, pair anthropometrics with metabolic markers (like fasting glucose, triglycerides, and inflammatory markers) to get a fuller picture.
Are there medical red flags that suggest adipose tissue growth is failing?
Signs that expansion is becoming dysfunctional include increasing fibrosis and chronic low-grade inflammation patterns (for example, persistently elevated CRP or adipokine imbalance), plus evidence of ectopic fat deposition such as elevated liver fat or insulin resistance that does not improve with lifestyle changes. If these patterns are present, it is worth discussing evaluation with a clinician rather than assuming it is only lifestyle-related.
Which Epidermis Cells Grow and Divide
Epidermis growth comes from basal stratum basale keratinocyte stem and progenitor cells that mitose and replace cells ou