The short answer: the cells in the epidermis that grow and divide are the keratinocytes in the stratum basale, the deepest epidermal layer. Specifically, it is the keratinocyte stem cells and their progenitor (transient amplifying) cells that run mitosis continuously, keeping your skin renewed from the inside out. Every other layer above the basale is largely a post-division story: cells differentiating, flattening, and eventually shedding off the surface.
Which Epidermis Cells Grow and Divide
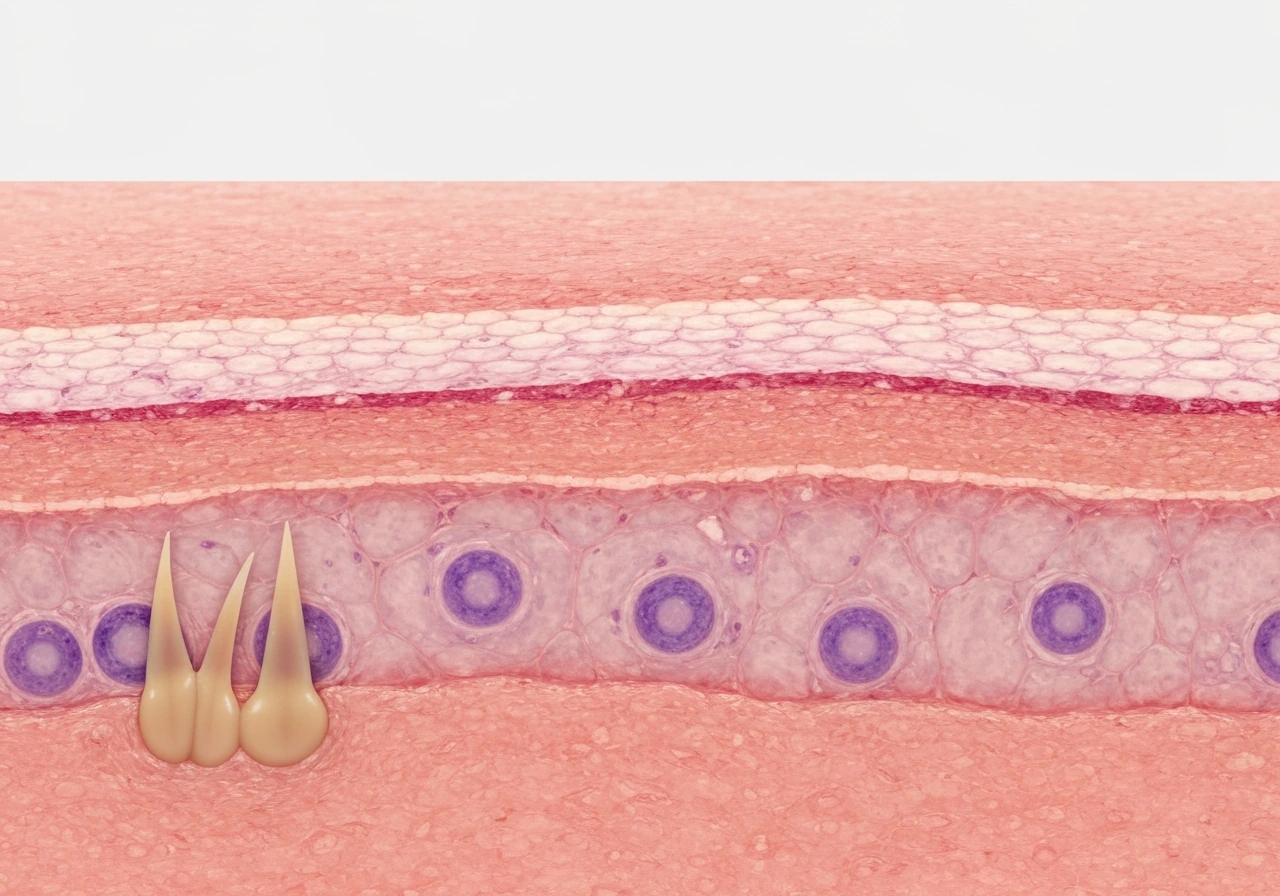
A quick layer-by-layer map of the epidermis
Before diving into which cells divide, it helps to picture the epidermis as a four-floor building where ground-floor tenants are the workers and upper-floor residents are slowly packing up to leave. From deep to surface, the four main strata are:
| Layer | Alternate name | Main cell activity | Key markers |
|---|---|---|---|
| Stratum basale | Basal layer | Mitosis / active division | K5, K14 keratins |
| Stratum spinosum | Spinous layer | Early differentiation, limited division | K1, K10 keratins begin appearing |
| Stratum granulosum | Granular layer | Late differentiation, keratohyalin granules form | Profilaggrin → filaggrin, involucrin |
| Stratum corneum | Cornified layer | Dead, anucleate corneocytes; barrier function | Loricrin, cornified envelope proteins |
The stratum basale sits right on top of the basement membrane, anchoring to the dermis beneath it. Moving upward, each layer represents a further stage of a journey that ends with dead, protein-packed cells flaking off your skin surface every day. Understanding how epithelial tissue grows puts this layered progression in broader context: the epidermis is just one spectacular example of a self-renewing epithelium.
The direct answer: basal keratinocytes are the dividers
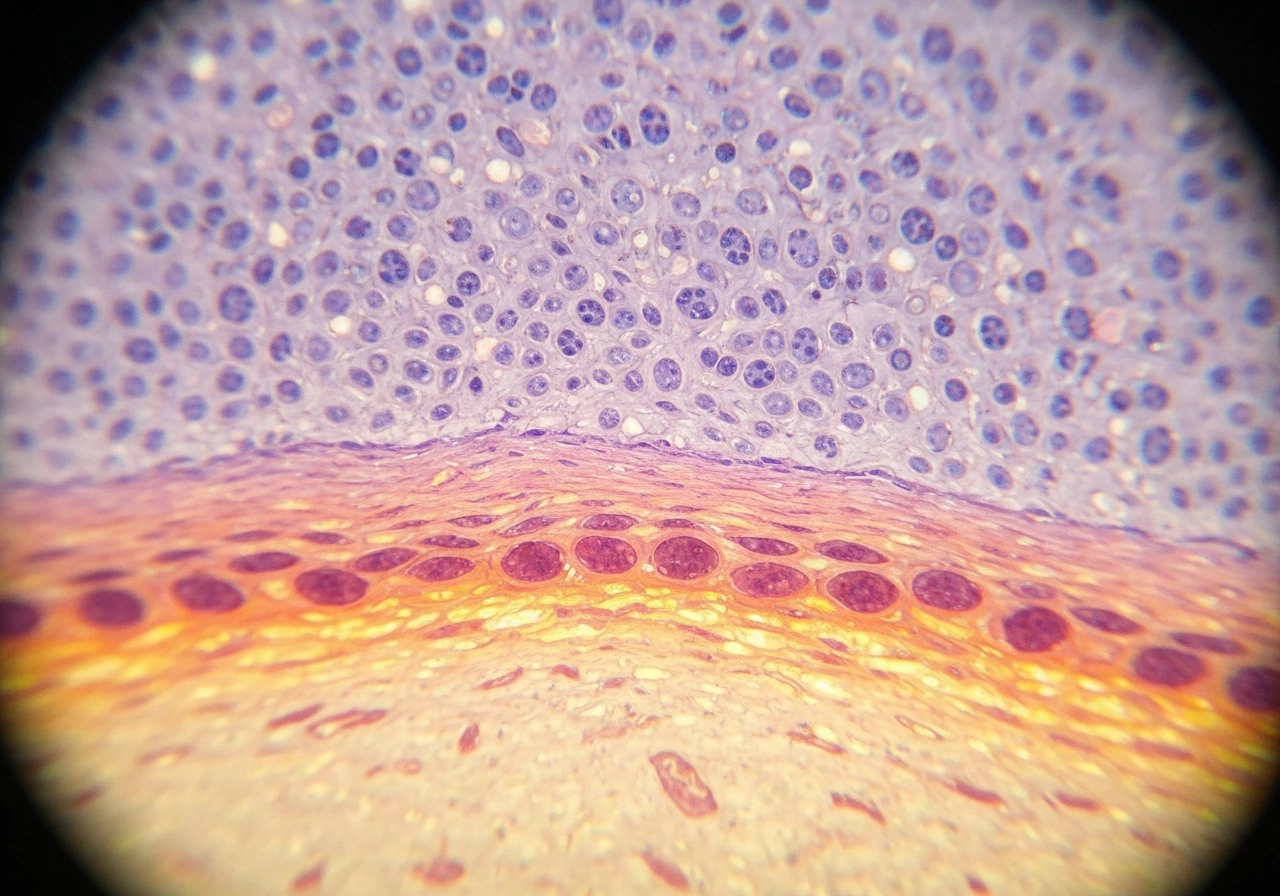
The cells doing the actual growing and dividing are the basal keratinocytes sitting in the stratum basale. This population contains two types you should know: stem cells, which divide slowly and indefinitely (they are the long-term replenishment source), and transient amplifying (TA) cells, which are the stem cells' immediate daughters. TA cells divide rapidly several times before committing to differentiation. Together, these two cell types are responsible for all the mitotic activity that keeps the epidermis intact.
These basal cells are identifiable by the keratins they express: K5 and K14. Think of K5/K14 as the molecular uniform of a proliferating basal keratinocyte. The moment a cell commits to moving upward and differentiating, it switches its keratin expression to K1 and K10, which is a reliable molecular signal that division is essentially done. If you ever see an exam question asking which protein markers indicate an actively dividing epidermal cell, K5 and K14 are your answer.
It is worth asking: why do cells grow and divide in the first place? For basal keratinocytes, the drive is tissue maintenance. Skin loses roughly 30,000 to 40,000 surface cells every hour, so the basale has to keep producing replacements at pace.
Where division does NOT happen: the suprabasal layers
Once a keratinocyte leaves the stratum basale and enters the stratum spinosum, active division largely stops. The spinosum gets its name from the spiny appearance of its cells under a microscope, caused by desmosomes (protein bridges) holding neighboring cells together. There is some evidence that the lower spinosum retains a tiny amount of proliferative capacity, but it is minimal compared to the basale, and for practical purposes you can treat the spinosum as the beginning of the differentiation zone.
By the time cells reach the stratum granulosum, differentiation is in full swing. These cells are packed with keratohyalin granules containing profilaggrin, a precursor protein that gets processed into filaggrin. Filaggrin bundles keratin filaments together and is a critical part of building the cornified envelope, the structural scaffold that makes the outer skin barrier so tough. No meaningful mitosis is happening here. Cells in the granulosum are preparing to die in a controlled, programmed way.
The stratum corneum is the final stop. These are anucleate corneocytes, essentially protein-filled sacs locked into a lipid matrix. They are dead by every biological measure and serve purely as a physical and chemical barrier. Understanding this gradient is what separates a beginner's answer from a solid one: it is not that suprabasal cells cannot divide, it is that differentiation and division are mutually exclusive programs, and once the differentiation switch flips, division is off.
How epidermal growth actually works over time
Think of the epidermis like a conveyor belt running from the basement membrane to the skin surface. Basal keratinocytes divide, and one daughter cell stays behind as a stem or progenitor cell while the other gets pushed upward. That upward cell then differentiates progressively through each layer, changing its protein expression and internal architecture along the way, until it flattens into a corneocyte and eventually sheds. The entire transit time from basal division to surface shedding is roughly 2 to 4 weeks in normal human skin.
This is a neat example of the two processes by which tissues grow: cell proliferation and cell enlargement. In the epidermis, proliferation happens in the basale, while differentiation-driven changes in cell size and shape happen across the suprabasal layers. Both processes contribute to the tissue's overall architecture, just at different locations.
This migration-and-differentiation cycle also explains why skin conditions like psoriasis (where basal cell division becomes abnormally rapid) lead to thickened, scaly skin. When the conveyor belt speeds up without the differentiation program keeping pace, you get immature cells at the surface and a disrupted barrier. The biology of the basale is not just exam material. It explains real diseases.
Stem cells vs. transient amplifying cells: why both matter
Basal keratinocyte stem cells are slow-cycling and long-lived, designed for decades of skin maintenance. Their daughters, the transient amplifying cells, are the sprint workers: they divide several times quickly, then exit the basale and differentiate. This two-tier system is efficient because it limits the number of times stem cell DNA has to be copied (reducing mutation risk) while still generating large numbers of replacement cells fast. Do cells grow at different rates depending on their role? Absolutely, and the epidermis is a textbook example of that principle in action.
How scientists confirm which cells are dividing

You might wonder: how do researchers actually know it is the basal layer doing the dividing? A few methods confirm this reliably. The most classic approach is staining tissue sections for mitotic figures (cells caught mid-mitosis under a light microscope). You will see these almost exclusively in the stratum basale. More modern approaches use Ki-67 immunostaining, a protein expressed only during active cell-cycle phases (G1, S, G2, and M). Ki-67-positive cells in skin sections cluster almost entirely in the basale.
Another approach involves pulse-labeling with BrdU (bromodeoxyuridine), a synthetic nucleoside that incorporates into newly synthesized DNA during S phase. After a short pulse, BrdU-labeled cells appear only in the basal layer. After a longer chase period, labeled cells appear progressively higher in the spinosum and granulosum, neatly tracking the migration of post-mitotic cells upward through the tissue. This kind of experiment is what put the cell-division story in the basale on solid scientific footing.
If you want to understand how a cell grows before it divides, the basal keratinocyte is a great case study: it grows to a threshold size, commits to mitosis, and then produces daughters that either stay in the basale or move upward. The cell-cycle checkpoints governing this are the same G1/S/G2/M framework you study in any intro biology course, just applied to a tissue with a very clear spatial organization.
Keratin markers as a practical diagnostic tool
Because K5 and K14 label basal (proliferative) cells while K1 and K10 label suprabasal (differentiating) cells, pathologists use these markers routinely. A skin biopsy showing K5/K14 expression in upper layers, where it does not belong, suggests an abnormal proliferative state. Conversely, missing K1/K10 in the spinosum might point to a differentiation defect. How cells grow or increase in size is only part of the story; what they express as they grow tells you everything about whether the tissue is behaving normally.
Comparing the epidermal layers at a glance
| Layer | Divides? | Differentiates? | Key proteins | Fate |
|---|---|---|---|---|
| Stratum basale | Yes (actively) | No (uncommitted) | K5, K14 | Stays or moves up |
| Stratum spinosum | Rarely / minimally | Yes (early) | K1, K10, desmoglein | Migrates upward |
| Stratum granulosum | No | Yes (late) | Filaggrin, involucrin, loricrin | Undergoes cornification |
| Stratum corneum | No | Complete | Cornified envelope proteins | Shed from surface |
The contrast between the basale and the corneum is about as stark as biology gets: one layer is a nursery of dividing, nucleus-bearing cells; the other is a graveyard of protein envelopes doing their final job as a barrier. Do cells get bigger as you grow? In the epidermis the answer depends entirely on which layer you are looking at, and that distinction matters enormously for understanding how skin functions.
What about sebaceous glands and hair follicles?
A quick note for curious readers: the epidermis is not the only place skin cells divide. Hair follicle bulge cells and sebaceous gland progenitors also undergo mitosis, and the follicle bulge is actually considered a reservoir of epidermal stem cells that can repopulate the interfollicular epidermis after injury. So when someone asks specifically about interfollicular epidermis, the answer is unambiguously the stratum basale. When skin is wounded or a follicle is involved, the story gets a bit wider. How adipose tissue grows shows a useful contrast: fat tissue grows primarily by cell enlargement and by adding new adipocytes from preadipocyte progenitors, a fundamentally different strategy from the rapid turnover and surface-shedding you see in skin.
What to remember for exams (and real life)
If you are studying for a biology or anatomy exam, here is the distilled version of everything above:
- The stratum basale is the only epidermal layer with significant mitotic activity. This is the correct answer to 'which cells in the epidermis grow and divide.'
- The dividing cells are keratinocyte stem cells and transient amplifying cells. Both are in the basale.
- K5 and K14 keratins = basal, proliferative. K1 and K10 keratins = suprabasal, differentiating. These molecular switches are widely tested.
- Suprabasal layers (spinosum, granulosum, corneum) are differentiation and migration zones, not division zones.
- Filaggrin (processed from profilaggrin in the granulosum) and cornified envelope proteins (involucrin, loricrin) mark late differentiation and cornification. They appear well above the dividing cells.
- Ki-67 staining and BrdU pulse-chase experiments both confirm that mitosis is confined to the basale in normal skin.
- The full transit from basal division to surface shedding takes roughly 2 to 4 weeks.
The core concept here is a stem-cell-driven conveyor belt: cells are born in the basale, differentiate as they travel upward, and shed from the surface. Keep that image in mind and you will be able to reason through almost any epidermis question, whether it is about which layer expresses a given protein, why a drug targeting rapidly dividing cells damages skin, or why wound healing can take weeks. The basale is where it all starts.
FAQ
Do suprabasal (stratum spinosum/granulosum) keratinocytes ever divide?
Under normal conditions, “which cells in the epidermis grow and divide” means the keratinocytes in the stratum basale. In most textbook cases, you will not count suprabasal keratinocytes as meaningfully proliferative because once a cell initiates differentiation it shuts down the division program. The key exception is after injury, where local proliferation can expand beyond the usual basale pattern to help restore the barrier.
Why might I see mitosis in layers other than the stratum basale during a wound or healing response?
Not at baseline, but after burns or chronic irritation the epidermis can ramp up cell cycling. In wound healing, basal cells and nearby progenitors increase proliferation to refill epidermal layers, so mitotic activity can be temporarily observed outside the typical basale “stripe.” The takeaway is that the dividing compartment is usually basale, but it can widen during repair.
How does the answer change if the question is about interfollicular epidermis versus hair follicles?
Cell division is typically described for the interfollicular epidermis. Hair follicle bulge keratinocyte stem cells and sebaceous gland progenitors can also divide, but they are not part of the interfollicular epidermis layer stack you are usually tested on. If an exam specifies “interfollicular epidermis,” the answer remains stratum basale.
What should I conclude if K1/K10 are missing in the suprabasal layers?
The epidermis is organized so that differentiation switches off the cell cycle. Practically, this means you should not expect markers of active proliferation (for example, K5 and K14, or Ki-67 positivity) to persist in the upper layers. If upper layers show proliferative markers, it suggests disease or a failed differentiation program.
Can keratin markers (K5/K14 versus K1/K10) tell me whether a skin sample is proliferating even if Ki-67 is not measured?
K5 and K14 are associated with the proliferative basal state, while K1 and K10 track keratinocyte differentiation. In some skin conditions, the “marker switch” is uncoupled, so keratin expression can become abnormal even if division is not maximal everywhere. That is why pathology uses both proliferation and differentiation markers together, not just one set.
Why can’t cells in the stratum corneum divide?
Yes, corneocytes are dead and anucleate, so they cannot divide. Division requires DNA replication and mitotic machinery, which corneocytes do not have. They are part of the barrier, not the renewal compartment.
Is the 2 to 4 week turnover time the same for everyone?
Transit time from basale division to surface shedding is often cited as about 2 to 4 weeks in healthy adult skin, but it can change with factors like age, body site, and conditions that alter turnover. If you are analyzing a real biopsy or clinical timeline, assume turnover is variable and do not treat the 2 to 4 week window as a fixed constant.
Why might a basal keratinocyte not show Ki-67 staining but still be part of the stem/progenitor pool?
Because proliferation is tied to cell-cycle state, a basal cell can be “Ki-67 negative” if it is temporarily out of active phases, even though it still belongs to the basal compartment. Conversely, Ki-67 positive staining identifies cells currently in an active cycle range. So timing and marker interpretation matter when you compare samples.
What are common reasons exam or lab results might suggest dividing cells are not in the basale even when they are?
Biopsies can show a regional pattern, especially in diseased or scarred skin, where turnover and differentiation can be spatially uneven. Sampling at the wrong depth or at an atypical region can make it look like dividing cells are “missing” or shifted. For interpretation, confirm layer orientation and compare with adjacent normal or peri-lesional tissue when possible.
Why Do Cells Grow? Signals, Cell Cycle, and Size Limits
Why cells grow: signals trigger biomass and cell cycle readiness, but physics, energy, and DNA limits cap size.