Yes, stem cells can grow cartilage, here's what that actually means
The short answer is yes. Stem cells can be coaxed into becoming cartilage-producing cells called chondrocytes, and under the right conditions they will build actual cartilage tissue around themselves. This has been demonstrated repeatedly in labs, in animal models, and in a growing number of early human clinical trials. But the longer answer matters a lot, because "growing cartilage" can mean three very different things: producing cartilage cells in a dish, engineering a cartilage-like tissue construct in the lab, or successfully regenerating cartilage inside a living joint. Each of those has different requirements, different success rates, and different timelines. If you're trying to figure out whether this is a real option for you today, you need to understand which of those three things is actually being offered.
Which stem cell types can actually become cartilage

Not all stem cells are equally good at making cartilage, and this is where differentiation, the process of a stem cell committing to a specific cell identity, really matters. Think of it like a student choosing a major: the earlier they are in their degree, the more flexibility they have, but the harder it is to get them to commit to one path without wandering off into something else.
Mesenchymal stem cells (MSCs), sometimes called mesenchymal stromal cells, are the current workhorse for cartilage regeneration. They naturally sit in bone marrow, fat tissue, and synovial fluid (the lubricating fluid in your joints), and they have a strong natural tendency to differentiate into cartilage, bone, and fat cells. Because they're adult stem cells, they're also less likely to form tumors, which makes them more clinically practical right now. If you want to understand where stem cells grow in the body and how that location shapes their potential, MSCs are a perfect example: their tissue of origin directly influences their differentiation bias.
Induced pluripotent stem cells (iPSCs) and embryonic stem cells (hESCs) are pluripotent, meaning they can theoretically become any cell type in the body. That sounds like a huge advantage, and in some ways it is, but pluripotency also means these cells require more precise differentiation protocols to reliably produce chondrocytes rather than veering into other tissue types. The International Society for Stem Cell Research (ISSCR) specifically flags tumorigenicity risk as a major concern for pluripotent stem cell-based interventions, because any undifferentiated cells left in the final product could potentially form tumors called teratomas. Stringent purity and differentiation controls are non-negotiable before these cells go anywhere near a patient.
| Stem Cell Type | Source | Chondrogenic Potential | Tumor Risk | Clinical Status |
|---|
| Mesenchymal Stem Cells (MSCs) | Bone marrow, fat, synovium | High, natural tendency | Low | In clinical trials and some approved therapies |
| Induced Pluripotent Stem Cells (iPSCs) | Reprogrammed adult cells | High with correct protocol | Moderate to high (requires tight control) | Mostly preclinical/early trials |
| Embryonic Stem Cells (hESCs) | Early human embryos | High under hypoxic conditions | Moderate to high | Early clinical research stage |
| Adipose-Derived Stem Cells (ADSCs) | Fat tissue | Moderate | Low | In several clinical trials |
The bottom line on cell type: MSCs are the practical frontrunner today. iPSCs and hESCs are the more powerful but more complex option being refined for the future. It's also worth noting that growing stem cells in sufficient quantities and keeping them stable is itself a significant challenge that researchers are still actively solving.
How cartilage is actually "grown", lab culture vs. tissue engineering
There are two broad approaches to growing cartilage from stem cells, and they work very differently.
The first is cell-based therapy, where stem cells are expanded in culture, differentiated into chondrocytes (or chondrogenic progenitors), and then injected or transplanted directly into a damaged area. The idea is that once in the right environment, the cells will continue differentiating and secreting the extracellular matrix (collagen type II, aggrecan, glycosaminoglycans) that makes real cartilage springy and load-bearing. This is simpler to manufacture and deliver, but it gives you less control over what the cells do after implantation.
The second approach is tissue engineering, where you build a cartilage construct outside the body first and then implant it. Stem cells are seeded onto a scaffold (more on that shortly), given chondrogenic signals, and cultured for weeks in a bioreactor until they've deposited a cartilage-like matrix. The result is a 3D tissue you can characterize and quality-check before it ever enters the patient. This is more complex to manufacture but gives you far more control over the final product. The FDA's 2023 draft guidance on potency assurance for cellular and gene therapy products is directly relevant here: it recommends science- and risk-based potency strategies during manufacturing, which means showing that your product actually does what you claim it does before it reaches a patient.
The signals that tell stem cells to become cartilage
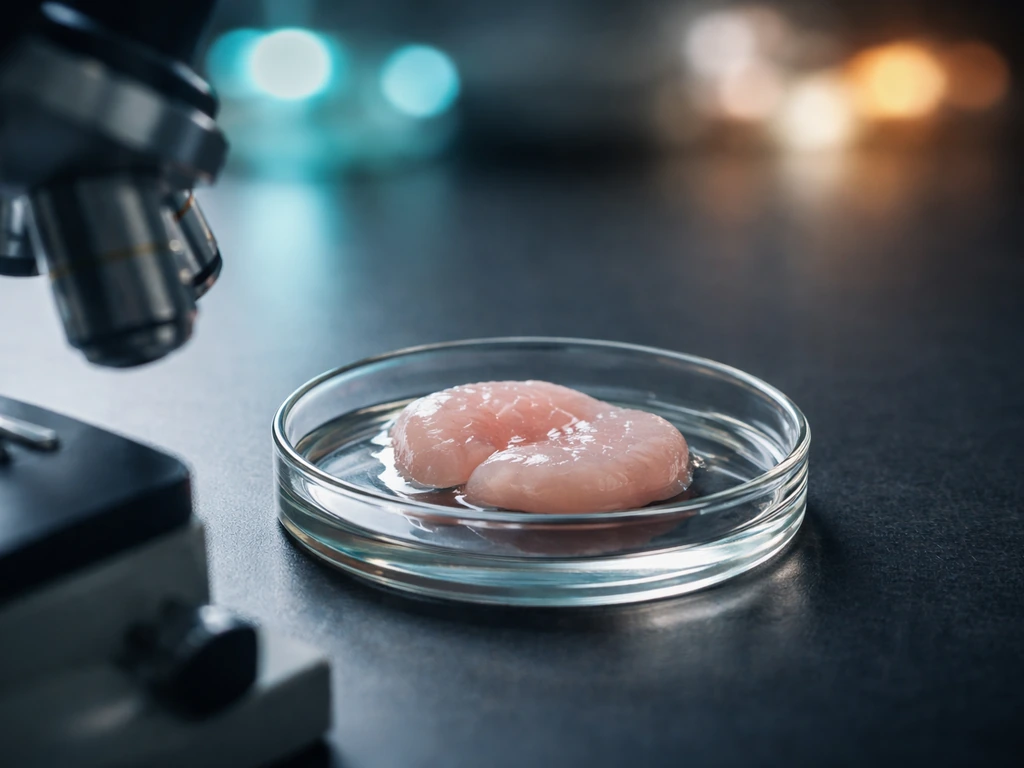
Chondrogenesis, the process of making cartilage, doesn't happen automatically just because you put stem cells in a joint. It requires a specific cocktail of molecular signals, and getting this right is where most of the scientific work is concentrated.
The most important players are transforming growth factor-beta (TGF-β), particularly TGF-β1 and TGF-β3, which are the primary drivers of chondrogenic differentiation in MSCs. Bone morphogenetic proteins (BMPs), especially BMP-2, BMP-4, and BMP-6, work alongside TGF-β to push cells further down the cartilage pathway. Insulin-like growth factor 1 (IGF-1) helps maintain the cartilage phenotype and promotes matrix production. A transcription factor called SOX9 sits at the center of the whole process: it's the master regulator that turns on the genes for collagen type II and aggrecan, the proteins that define real hyaline cartilage (as opposed to the weaker fibrocartilage that often forms in natural wound healing).
Oxygen tension is another variable that most people overlook, and it makes a measurable difference. Cartilage in the body is naturally avascular, it has no direct blood supply, which means it lives in a low-oxygen environment. Research on human embryonic stem cells found that chondrogenic differentiation under hypoxic conditions (around 2% oxygen, compared to the roughly 20% oxygen in normal air) enhanced cartilage protein synthesis and improved biomechanical performance of the resulting tissue. This makes biological sense: you're replicating the native environment that cartilage normally develops in. MSC-based models show the same effect, with hypoxia linked to higher glycosaminoglycan production and stronger expression of SOX9 and its downstream genes, partially driven through HIF (hypoxia-inducible factor) signaling pathways.
Why the culture environment matters so much
Beyond growth factors and oxygen, the physical format of the culture matters too. Chondrocytes are notoriously sensitive to their surroundings. When you culture them on a flat plastic dish in 2D, they tend to dedifferentiate, they lose their chondrocyte identity and start behaving more like fibroblasts (generic connective tissue cells). This is one of the core practical problems in cartilage cell therapy. Research into whether stem cells can grow on plastic surfaces gets directly at this issue: surface chemistry and dimensionality both influence how stem cells behave and what they differentiate into. Pellet culture (where cells are centrifuged into a small 3D ball) and aggregate culture models preserve chondrogenic identity far better than flat dishes, and that's one reason bioreactor-based tissue engineering has become the preferred approach for building functional cartilage constructs.
Scaffolds and the physical structure of cartilage growth

Cartilage isn't just cells, it's mostly extracellular matrix. About 70-80% of cartilage by wet weight is water, held in place by a network of collagen fibers and proteoglycans (large sugar-protein molecules like aggrecan). The cells only make up a small fraction. So when you're engineering cartilage, you need something that gives the cells a 3D environment to grow into and a template to organize their matrix production around. That's what scaffolds do.
Scaffolds come in several material categories, each with different trade-offs.
- Hydrogels (alginate, fibrin, hyaluronic acid, gelatin): These are water-swollen polymer networks that closely mimic the soft, hydrated environment of cartilage. They're good at maintaining chondrocyte phenotype and are often injectable, which makes them useful clinically. The downside is that they tend to be mechanically weak compared to real cartilage.
- Natural biopolymers (collagen, chitosan): These offer good cell compatibility and bioactive cues but can degrade unevenly and are harder to standardize in manufacturing.
- Synthetic polymers (PLGA, PCL, PLA): These are more mechanically tunable and predictable in degradation rate, but they don't provide the same biological signals that natural materials do. Cells sometimes need extra biochemical nudges to behave correctly on them.
- Decellularized extracellular matrix (dECM): This involves stripping cells from donor cartilage tissue and using the remaining matrix as a scaffold. It preserves the native architecture and biochemical cues beautifully, but sourcing and standardization are challenges.
Mechanical loading is another factor that's easy to underestimate. Cartilage in the body is constantly compressed, stretched, and sheared as you move. These physical forces aren't just something it has to withstand, they're actually part of what makes it develop and maintain itself. Bioreactors that apply cyclic compression or fluid shear to developing cartilage constructs consistently produce tissue with better mechanical properties than static cultures. This is a great example of a physical constraint on growth that turns out to be a requirement, not just a challenge: without load, you don't get properly organized collagen architecture.
Interestingly, the same principles of physical constraint and environmental signal govern how stem cells form bone, which shares a developmental lineage with cartilage. Work on whether stem cells can grow bone reveals overlapping mechanisms, and in fact one of the biggest problems in cartilage tissue engineering is preventing stem-cell-derived chondrocytes from hypertrophying (over-maturing) and mineralizing into bone-like tissue instead of staying as stable cartilage.
What outcomes you can realistically expect today
Here's where honesty is essential, because the gap between lab results and clinical outcomes is still significant.
What's working
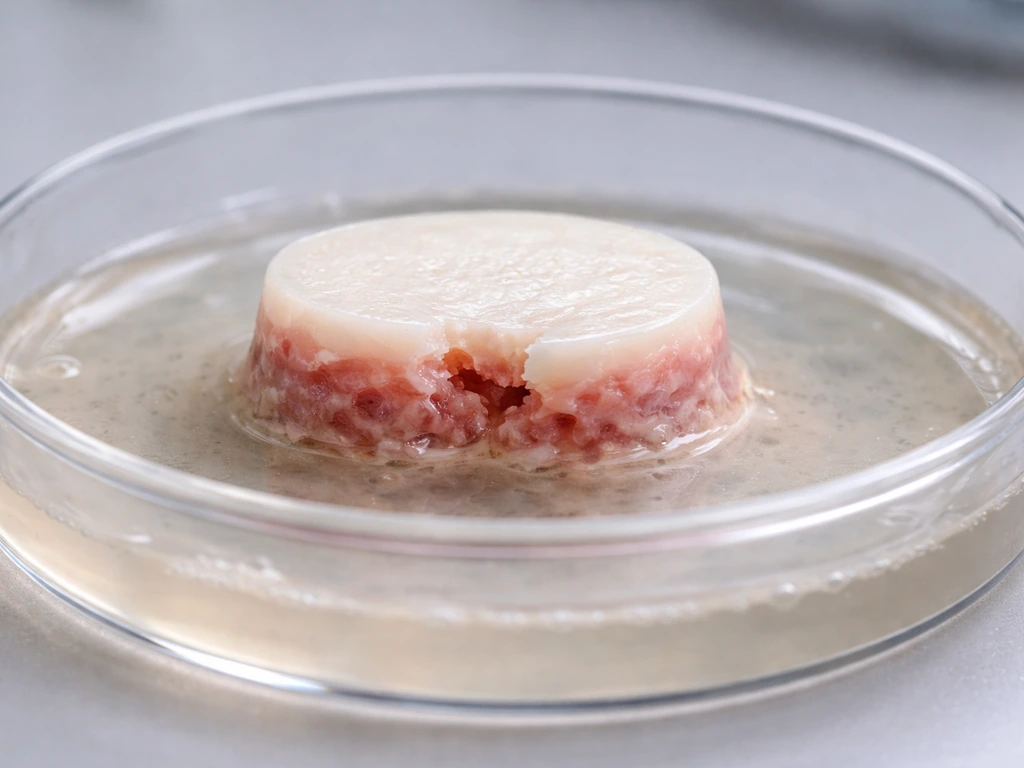
In research settings, stem cells (particularly MSCs) can reliably be differentiated into chondrocytes that produce type II collagen and glycosaminoglycans, the biochemical signature of hyaline cartilage. Tissue-engineered constructs have achieved mechanical properties in the range of native cartilage in some studies. Early clinical trials using autologous (your own) MSCs for knee cartilage defects have reported promising safety profiles and functional improvements in patient-reported outcomes. Some cell-based products for cartilage repair have received regulatory approval in certain markets (notably Korea and Japan), though the evidence base varies.
The real limitations
The main problems are integration, durability, and tissue quality. Even a well-made cartilage construct faces the challenge of bonding to the surrounding native cartilage, which, because it's avascular, has almost no natural repair machinery to help incorporate new tissue. Fibrocartilage (a weaker, scar-like version of cartilage) often forms instead of true hyaline cartilage, both in cell injection therapies and in some engineered constructs. Long-term durability data in humans is still limited because many of these approaches are less than a decade into clinical use.
Dedifferentiation during cell expansion is a practical manufacturing headache: the more you expand MSCs or chondrocytes to get enough cells for a therapy, the more they tend to lose their cartilage-forming identity. There are also immune considerations when using allogeneic (donor) cells, even MSCs, which are often described as immune-privileged, can trigger responses in some recipients. And for pluripotent stem cell-derived approaches, the residual tumor risk means extensive safety testing is required before clinical use.
How success is measured
- Biochemical markers: Collagen type II expression, glycosaminoglycan content, SOX9 activity — these indicate true chondrogenic identity rather than fibrocartilage formation.
- Mechanical properties: Compressive stiffness (Young's modulus) and dynamic mechanical analysis compared to native cartilage values (roughly 0.5-1.5 MPa in compression for articular cartilage).
- Histological integration: Whether the engineered tissue connects structurally to surrounding cartilage at the implant site.
- Clinical outcomes: Patient-reported pain and function scores, MRI assessment of defect fill, and long-term follow-up for durability.
Practical next steps if this matters to you personally

If you're considering a stem cell-based cartilage therapy for a joint problem, or evaluating this as a clinician or researcher, here's how to approach it practically.
- Find out exactly what type of stem cells are being used and where they come from. Autologous MSCs (from your own body) carry lower immune and tumor risk than allogeneic or pluripotent-derived cells. Ask specifically whether the cells are fully characterized and whether the product has regulatory clearance in your country.
- Ask what the differentiation protocol is and how chondrogenic commitment is verified. A credible provider should be able to tell you that they confirm SOX9 expression and collagen type II production before using the cells — not just that they "became chondrocytes."
- Ask about the scaffold or delivery system. Injecting bare cells is very different from implanting a matured cartilage construct. Know what you're actually receiving.
- Look for peer-reviewed clinical trial data, not just testimonials or case reports. Search ClinicalTrials.gov for registered trials using your condition and stem cell type. Registered trials follow standardized protocols and safety monitoring that individual clinics offering stem cell treatments often do not.
- Be skeptical of clinics operating outside regulatory frameworks. Legitimate cell therapy research follows FDA (in the US) or equivalent agency guidance on potency assurance and product characterization. If a provider can't explain their quality controls, that's a red flag.
- Ask about long-term follow-up. Cartilage is a long-term tissue. A therapy that looks good at 6 months but hasn't been tracked at 3-5 years is telling an incomplete story.
- Consult an orthopedic specialist or sports medicine physician familiar with cartilage repair options. Stem cell-based approaches exist alongside other options (microfracture, autologous chondrocyte implantation, osteochondral grafting) and the best choice depends on defect size, location, age, and activity level.
One more thing worth noting: the field of cartilage tissue engineering is genuinely moving fast. What was only possible in a research lab five years ago is now entering early clinical practice. Staying current means looking at sources like the ISSCR's published guidelines and FDA guidance documents, not just news articles about breakthroughs. The mechanisms governing how stem cells grow, differentiate, and form tissues are becoming better understood every year, and the practical outcomes are improving alongside that understanding.