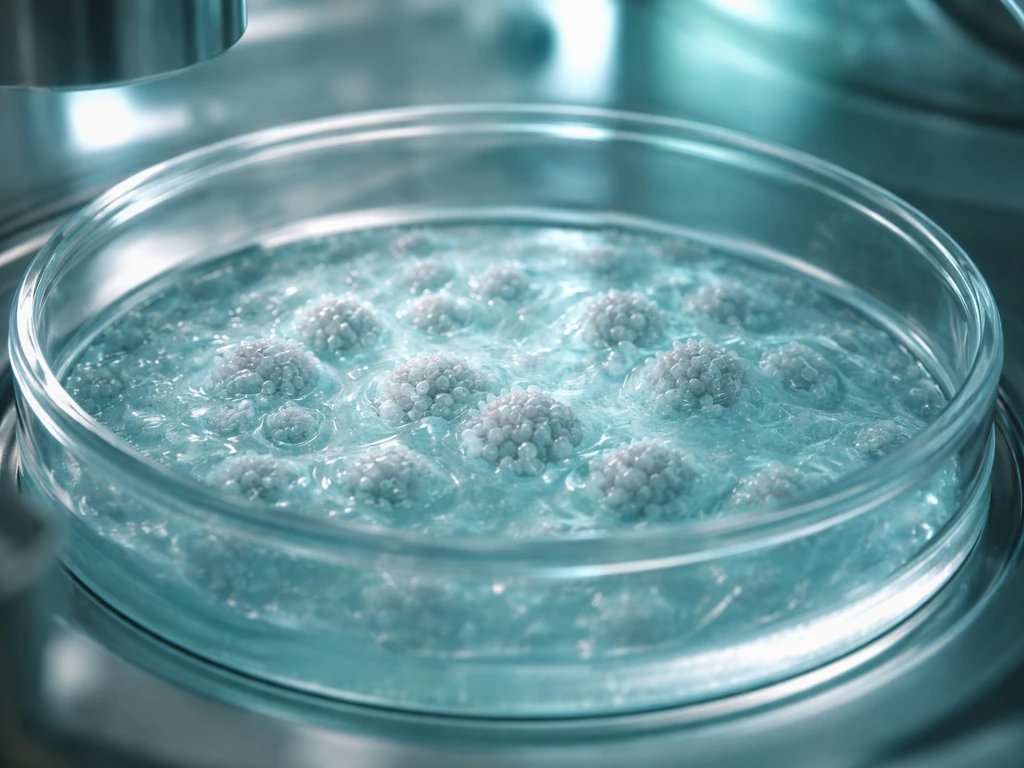
Yes, stem cells can be grown, but the word 'grown' means something very specific here, and whether you can do it depends almost entirely on who you are and what equipment you have access to. In a professional research or clinical lab, scientists grow stem cells routinely by culturing them in controlled dishes and flasks. Outside of that setting, the honest answer is no, at least not safely or legally. Let's break down exactly what's involved so you know what's realistic and what's not.
Can You Grow Stem Cells? What It Means and How It Works

What 'growing stem cells' actually means
When most people ask this question, they have one of two things in mind. The first is in vitro culturing, which means keeping stem cells alive and multiplying them in a lab dish using nutrient-rich liquid media, coatings, and tightly controlled environmental conditions. The goal is to increase the number of stem cells while making sure they stay stem cells, not accidentally becoming heart muscle or nerve tissue before you want them to. This is the scientific standard meaning of 'growing stem cells.'
The second meaning is in vivo growth, which refers to what happens inside a living body when stem-cell-based therapies are used to stimulate tissue repair or remodeling. In the body, stem cells grow in specialized niches such as the bone marrow, skin, and intestinal lining. Think of it like sending in a repair crew to rebuild damaged cartilage or bone. This is not something you can trigger in your own body at will; it's the domain of clinical medicine and carefully regulated research programs. Both meanings matter, but they involve completely different processes and levels of access.
Types of stem cells and why the type changes everything

Not all stem cells are created equal, and the type you're working with has a massive impact on how you grow them, how strictly regulated the work is, and what you can actually do with the results. There are three major categories that come up in most discussions.
| Type | Source | Potency | Regulation Level | Ease of Culture |
|---|---|---|---|---|
| Embryonic Stem Cells (ESCs) | Early human embryos | Pluripotent (can become almost any cell type) | Very high, ethically restricted | Difficult, highly specialized |
| Adult Stem Cells (ASCs) | Bone marrow, fat, blood, etc. | Multipotent (limited range of cell types) | Moderate | Moderately difficult |
| Induced Pluripotent Stem Cells (iPSCs) | Reprogrammed adult cells (skin, blood) | Pluripotent (similar to ESCs) | High but fewer ethical barriers | Technically demanding, improving |
Embryonic stem cells are the most powerful but carry the heaviest ethical and legal restrictions because obtaining them requires destroying a human embryo. Induced pluripotent stem cells sidestep that by reprogramming ordinary adult cells back into a stem-like state using specific genetic factors, which is a major reason they've become central to modern research. Adult stem cells like mesenchymal stem cells from bone marrow are already used in some approved therapies, and they're the type most often discussed in topics like growing bone or cartilage from stem cells.
How stem cells are actually maintained and expanded in a lab
Growing stem cells in a lab is less like gardening and more like running a very finicky life-support system. In vitro culture choices, including whether cells can grow on plastic surfaces, can strongly affect how reliably you expand them. Every variable matters. Here is the standard process a research lab follows to keep a stem cell culture alive and expanding.
- Choose and prepare the culture vessel: Tissue culture flasks or dishes are coated with a substrate (like Matrigel, fibronectin, or laminin) that mimics the natural extracellular matrix stem cells cling to in the body. Without this, many stem cell types simply won't attach or survive.
- Prepare the culture medium: Stem cells need a specifically formulated liquid medium containing basal nutrients (glucose, amino acids, vitamins), serum or serum-free supplements, and critical growth factors. For iPSCs, popular media like mTeSR1 or Essential 8 are designed to maintain pluripotency without triggering differentiation.
- Control the environment: Cells go into an incubator set to 37°C (body temperature) with 5% CO2 and around 95% humidity. Oxygen levels are also managed, often held at 5% O2 rather than room air's 21%, because low oxygen better mimics the niches where stem cells naturally reside in the body.
- Feed and passage regularly: Stem cells consume nutrients and produce waste quickly. Media is typically changed every one to two days. When cells reach about 70-80% confluency (covering most of the dish), they must be 'passaged,' meaning gently detached, diluted, and replated into new vessels to prevent overcrowding, which triggers differentiation or cell death.
- Monitor for quality: Researchers regularly check cells under a microscope for the correct colony morphology, run assays to confirm pluripotency markers (like OCT4, SOX2, NANOG for iPSCs), and test for contamination.
The conditions that keep stem cells growing and potent
What makes stem cell culture genuinely tricky is that the same conditions that encourage growth can also accidentally push cells to differentiate, meaning they start becoming a specialized cell type and lose their stem-cell identity. Think of it like trying to keep bread dough perfectly at the right temperature: too cold and nothing happens, too hot and you ruin it. The signals that control this balance fall into several categories.
Growth factors and signaling molecules

Proteins like bFGF (basic fibroblast growth factor) are essential for maintaining human pluripotent stem cells. Other signaling pathways, particularly Wnt, Notch, and BMP, must be either activated or suppressed depending on what you want the cells to do. Getting these signals wrong, even slightly, tells the cells to start specializing. For adult stem cells used to grow bone or cartilage, different factors like TGF-beta, BMP-2, or IGF-1 are used to direct growth along specific tissue lineages.
Scaffolds and physical structure
Cells don't just respond to chemical signals; they also respond to the physical texture and stiffness of what they're sitting on. A scaffold that mimics spongy cartilage tissue sends different mechanical cues than one mimicking dense bone. Researchers designing tissue engineering experiments, including those exploring how stem cells grow cartilage or bone, tune scaffold stiffness precisely to guide differentiation in the right direction.
Oxygen tension and CO2
Standard room air has about 21% oxygen, but most stem cell niches in the body are significantly more hypoxic, closer to 2-5% oxygen. Culturing cells at physiological oxygen levels has been shown to preserve stemness better and reduce genomic stress over long culture periods. CO2 controls the pH of the media, and most stem cell media are buffered to stay around pH 7.4, just like blood. Drift in either direction causes rapid cell stress.
What can go wrong: differentiation, limits, and contamination
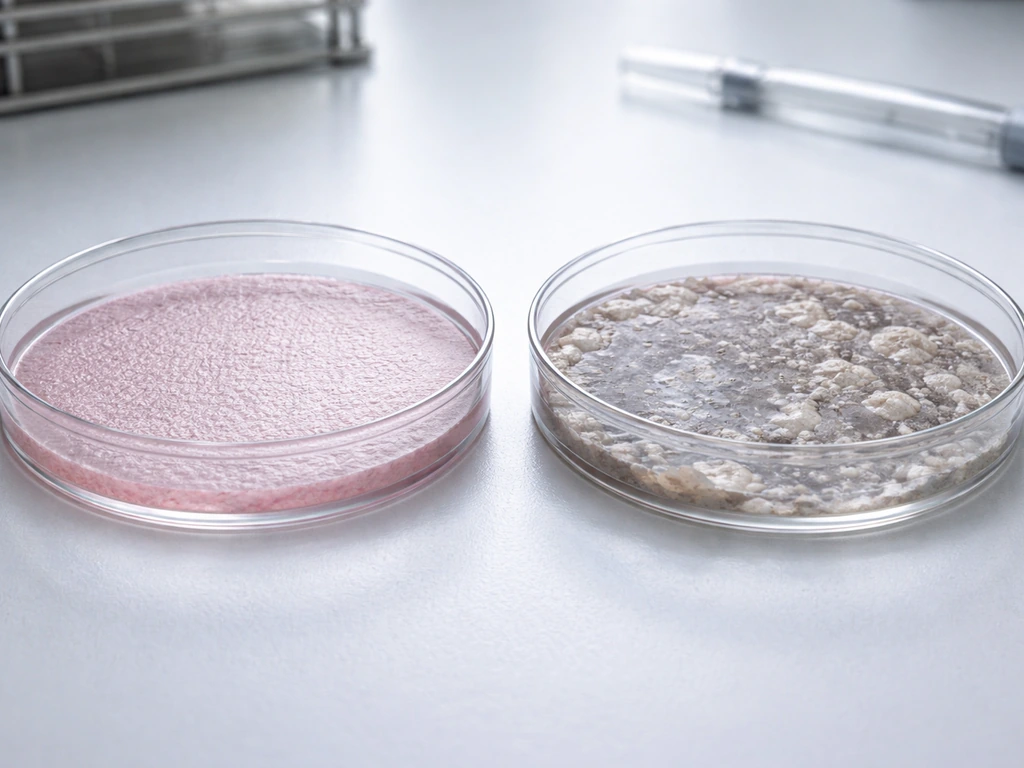
Even experienced researchers lose cultures. There are several major failure modes in stem cell growth, and understanding them explains why this can't just be improvised at home.
- Spontaneous differentiation: Without the right signals, stem cells start committing to a specific cell type on their own. Once differentiation begins, it's generally irreversible in that cell. You end up with a dish of specialized cells that no longer have the self-renewal properties you were trying to preserve.
- Replicative senescence: Stem cells don't divide forever. Adult stem cells especially have limited proliferative capacity. After enough passages, cells may slow down, stop dividing, or accumulate genetic errors. This is a fundamental biological constraint, not a fixable lab error.
- Mycoplasma and bacterial contamination: These are among the most common and devastating lab problems. Mycoplasma in particular is invisible under standard microscopes, has no obvious smell or color change in media, and can silently corrupt an entire culture over weeks before detection. Eliminating it requires specialized testing kits or PCR.
- Genomic instability: iPSCs and ESCs grown for extended periods can accumulate chromosomal abnormalities. This is a known safety concern for any clinical use of cultured stem cells and requires karyotyping to detect.
- Cross-contamination between cell lines: Labs working with multiple cell types risk accidentally mixing them, which has happened historically even in prestigious research institutions.
Can you grow stem cells at home? The honest answer
No, not safely, and in many contexts, not legally either. This isn't gatekeeping for its own sake; it comes down to genuine physical and regulatory barriers.
On the practical side: maintaining any mammalian cell culture requires a CO2 incubator (typically $3,000-$15,000 new), a biosafety cabinet to work sterile (another $2,000-$8,000), a cell counter, a quality microscope, a liquid nitrogen storage tank for cell banking, and a reliable supply of media and reagents that often require cold-chain shipping and have short shelf lives. That's before you account for the skill involved in sterile technique, which takes months to develop properly.
On the legal and ethical side: working with human embryonic stem cells is tightly regulated at national and institutional levels. In the United States, federal funding restrictions on ESC research have historically been significant, and working with human tissues generally requires IRB oversight, biosafety committee approval, and institutional licensing. Even adult stem cells derived from human donors carry HIPAA considerations and tissue banking regulations.
The risks of skipping this framework are not abstract. The FDA has issued repeated public safety alerts about unapproved stem cell and exosome products being marketed and administered outside of proper FDA review processes, warning that these can delay legitimate treatment and cause direct harm to patients. The International Society for Stem Cell Research (ISSCR) explicitly condemns administering unproven stem cell interventions outside of compliant clinical research or regulated medical innovation. If someone is offering you a way to grow or receive stem cells outside these structures, that is a red flag, not an opportunity.
Where to go from here if you're serious about this
There are legitimate pathways to get involved with stem cell science at every level of background and commitment. Here's how to think about your options today.
If you're a student or curious learner
Start with educational resources from organizations like the ISSCR, the NIH National Institute of Health's stem cell information pages, or university open courseware. Several universities and companies offer teaching kits using non-human cell lines (like yeast or plant cells) that let you experience cell culture techniques legally and safely at a fraction of the cost. These are genuinely good for building intuition about how cell growth works before ever touching mammalian cells.
If you want hands-on experience
Look for internship or volunteer opportunities at university research labs, hospital research departments, or biotech companies. Many academic labs welcome motivated undergraduates. Community biotech labs, sometimes called biohacker spaces, exist in many cities and often have trained staff, proper biosafety setups, and legal frameworks in place. Look for spaces registered with local biosafety authorities and ask directly what oversight they operate under.
If you're interested in stem cell therapy for a medical condition
The right path is through clinicaltrials.gov, where you can find FDA-registered trials investigating stem-cell-based treatments for specific conditions. Your doctor or a specialist can help you evaluate whether any trials are appropriate for your situation. Avoid any provider offering stem cell treatments outside of a clearly stated clinical trial or approved therapy framework.
Questions to ask before engaging with any stem cell program or provider
- Is this therapy or research approved or registered with the FDA (or your national equivalent)?
- Is the cell source and process fully disclosed, and has it been tested for safety in peer-reviewed research?
- What institutional oversight (IRB, biosafety committee) governs this work?
- Are results published in peer-reviewed journals?
- Are you being asked to pay out-of-pocket for experimental treatment? (This is a major red flag.)
Stem cells are genuinely one of the most fascinating growth systems in biology. They sit at the intersection of every major theme in how living things grow: cell division, signaling, environmental constraints, and the fine line between expansion and specialization. Whether you're curious about how stem cells grow bone, whether they can establish themselves on a scaffold, or simply how the body uses them to repair and renew tissue, the underlying biology rewards deeper study. The safest and most rewarding way in is through education and accredited institutions, not shortcuts.
FAQ
If I just want to grow stem cells for personal research, is there any safe way to do it at home?
No. Even if you use a non-embryonic line, expanding mammalian stem cells requires sterile technique, controlled incubator conditions, and quality testing to confirm the cells stayed stem-like. At home there is no reliable way to prevent contamination or verify cell identity, so it is not a safe or responsible setup.
Can I “grow” stem cells indirectly by taking a supplement or using a DIY protocol that stimulates my body?
What people call “growing” is usually marketing for signaling that may influence repair, but it is not the same as expanding stem cells in a controlled culture or directing clinically meaningful stem-cell behavior. Regulated options for in vivo stem-cell therapies go through trial design, dosing, and safety monitoring, so DIY approaches are high-risk and not equivalent.
Is it possible to start with induced pluripotent stem cells (iPSCs) and grow them without a lab?
Even iPSCs require the same core lab infrastructure as other stem-cell culture, including an incubator with correct oxygen and CO2, defined media, specialized growth matrices, and frequent passaging schedules. iPSC work also demands confirmatory assays to ensure reprogramming quality and genomic stability.
What’s the biggest sign that a stem cell provider is not legitimate?
A major red flag is offering treatment outside a clearly described clinical trial or approved therapy pathway, especially if they cannot explain the regulatory status, cell source, intended mechanism, and monitoring plan. Avoid anyone promising results, rapid “custom” growth, or charging fees that do not match a trial enrollment process.
If I join a clinical trial, will they let me keep the cells I’m given or take samples home?
Usually not. Many protocols collect and process cells under regulated handling rules, and participants typically do not receive cultured or modified materials for home storage or use. Trial staff can clarify what, if any, samples can be shared under consent and safety requirements.
Why do cultures sometimes stop growing even when the lab seems to follow the protocol?
Common causes include cells differentiating due to small signaling or timing errors, microbial or mycoplasma contamination that is hard to spot visually, and loss of viability from temperature or handling delays during passaging. Labs also track passage number because stemness and growth capacity can decline over time.
What does “stemness” mean in practice, and how is it checked?
Stemness refers to the cells maintaining their ability to behave like stem cells rather than committing to a specialized lineage. Researchers verify this with a combination of morphology, surface marker or gene-expression checks, and functional assays, not just observation of increased cell numbers.
Are there legitimate educational options that mimic mammalian stem-cell culture without using human cells?
Yes. Many teaching kits use non-human cell lines, or even non-mammalian models like yeast or plant cells, to demonstrate core ideas like sterility, growth curves, and passaging. These do not produce human stem cells, but they still build practical intuition for how culture conditions affect growth.
Can Stem Cells Grow Cartilage? How It Works and Limits
Learn if stem cells can grow cartilage, how chondrocytes form, and the limits, risks, and next steps to spot real progre