Yes, stem cells can grow bone. Certain stem cells, particularly mesenchymal stem cells (MSCs), have a well-documented ability to differentiate into osteoblasts, the cells that build bone tissue. When given the right biochemical signals and physical environment, those osteoblasts produce the collagen framework and mineral deposits that become real, functional bone. This has been shown in labs, in animal models, and in limited clinical settings. The catch is that "can" and "will" are two very different things, and the conditions required are precise, the clinical evidence is still maturing, and a lot of what gets marketed as stem-cell bone therapy is far ahead of where the science actually stands.
Can Stem Cells Grow Bone? How It Works and What’s Real

How stem cells fit into bone development
Bone is not a static material. It is living tissue that grows, remodels, and repairs itself throughout your life. The cells responsible for building new bone are called osteoblasts, and they originate from a precursor population of multipotent mesenchymal stem cells. Think of MSCs as uncommitted workers who can be assigned to different construction jobs: bone, cartilage, fat, or muscle, depending on the signals they receive. That flexibility is what makes them so interesting to researchers and clinicians.
In normal bone development, this process is tightly regulated from the moment an embryo starts forming a skeleton. MSCs in bone tissue reside in the periosteum (the outer membrane of bone) and in bone marrow. When you fracture a bone, a repair signal goes out, MSCs near the injury site wake up, start dividing, and a portion of them commit to the osteoblast lineage and begin laying down new bone matrix. Stem cells are not doing something exotic here. They are the ordinary biological machinery your body already uses.
How stem cells actually become bone: the differentiation pathway
The word that matters here is differentiation. A stem cell does not become bone on its own. It needs to receive the right molecular instructions that flip specific genetic switches, locking it onto the osteoblast path. Two master transcription factors sit at the top of that pathway: Runx2 and Sp7 (also called Osterix). Runx2 directs a multipotent mesenchymal cell to start committing to the osteoblast lineage, and Osterix acts downstream of Runx2 to complete that commitment. Mouse embryos with either gene knocked out produce no osteoblasts and therefore no bone at all, which tells you just how central these two factors are.
As differentiation progresses, you can track where a cell is in that journey by looking at what proteins it expresses. Early-stage osteoblast precursors show elevated alkaline phosphatase (ALP), bone sialoprotein (BSP), and type I collagen. Later, as the cells mature and begin mineralizing the matrix, osteocalcin (OCN) appears. Researchers use stains like alizarin red or calcein to visually confirm that real mineralization is happening, not just marker expression. This distinction matters: a cell can look like an osteoblast on paper (based on gene expression) without actually building mineralized bone. The gold standard is observing bone formation in a living system, with histology confirming lamellar collagen organization and mineralized matrix produced by human-origin cells.
The key signaling molecules that drive this process include BMP2 (bone morphogenetic protein 2), which pushes MSCs toward the osteoblast fate via Runx2 and Osterix, along with Wnt/beta-catenin signaling, which acts later to drive precursors further toward mature, bone-secreting osteoblasts. Indian hedgehog (Ihh) signaling works even earlier, initiating the bone formation program. These are not switches you can just flip manually; they interact in layered feedback networks that researchers are still mapping.
What stem-cell-derived bone actually needs to grow
Getting stem cells to commit to bone is only half the problem. Getting them to build functional bone tissue requires a supportive environment, and this is where most approaches live or die. Because growth on plastic depends heavily on the culture surface, researchers often compare stem-cell behavior on tissue-culture plastics versus more bone-relevant materials can stem cells grow on plastic. There are three core requirements: the right chemical signals (osteoinduction), a physical structure to grow on (osteoconduction), and a blood supply to keep the new tissue alive (vascularization).
Chemical signals and osteoinductive cues

In laboratory differentiation protocols, researchers typically expose MSCs or iPSC-derived progenitors to a cocktail of osteoinductive factors: BMP4, basic fibroblast growth factor (bFGF), dexamethasone, and ascorbic acid phosphate are common components. The exact schedule and concentration matter. One iPSC study found that which differentiation media protocol was used determined whether true bone formed in vivo at all. Cells differentiated with one protocol produced bone in transplanted animals; cells from a different protocol did not, even though both groups showed osteogenic markers in culture. This is a key lesson: marker expression in a dish does not guarantee bone formation in a body.
Scaffolds and physical support
Bone cells need something to anchor to and grow along. In tissue engineering, this is provided by scaffolds, porous three-dimensional structures made from materials like hydroxyapatite/tricalcium phosphate (HA/TCP), polycaprolactone (PCL), or composite materials like MgO/PCL. Scaffold design is not a minor detail. Pore size directly affects outcome: pores smaller than 100 micrometers can result in non-mineralized osteoid or fibrous tissue rather than proper bone. Macroporosity above 50 micrometers is important for cell colonization and for allowing blood vessels to grow in. Scaffold porosity, pore shape, and interconnectivity all need to be optimized to support cell viability, proliferation, and eventual mineralization.
Vascularization: the growth-limiting factor
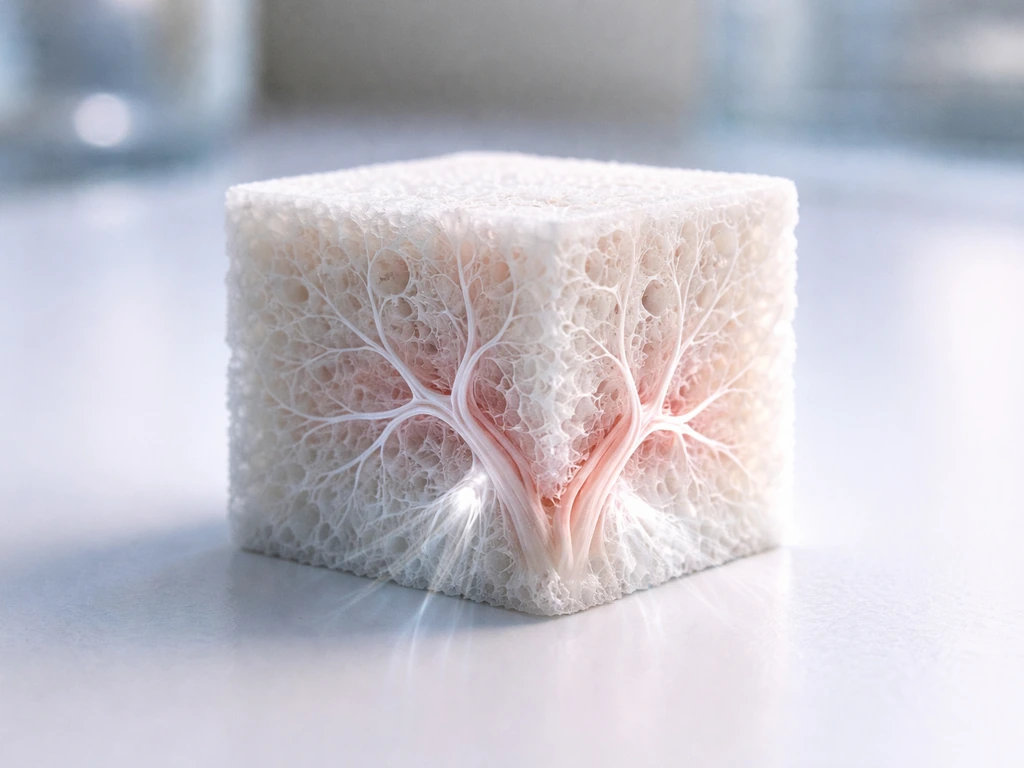
This is the wall that most bone tissue engineering approaches run into. New bone tissue needs oxygen and nutrients delivered by blood vessels. In the body, small bone grafts (a few millimeters thick) can be vascularized by ingrowth from surrounding tissue. But as the engineered bone construct gets larger, the interior cells starve before vessels can reach them. This is not unique to bone. It is a fundamental size constraint that applies to any dense tissue grown outside the body, and it is one of the main reasons lab-grown bone remains small or flat and why scaling up to, say, regenerating an entire vertebra outside the body is still largely unsolved.
Lab vs real life: where does stem-cell bone growth actually happen?
The science is genuinely impressive in controlled settings. Human embryonic stem cell (hESC) progeny have been shown to form histologically confirmed bone of human origin in animal transplant models, with evidence of mineralized matrix and lamellar collagen at 8, 12, and 16 weeks after transplantation. Human-origin cells were confirmed using ALU repetitive DNA in situ hybridization, which detects human-specific DNA sequences, so this was not just wishful interpretation. iPSC-derived cells have similarly produced true in vivo bone when the right differentiation protocol was used. MSCs derived from placenta and other sources have shown ectopic bone formation after transplantation in immunodeficient mice using HA/TCP particles as a carrier vehicle. These are real results in controlled research environments.
The clinical picture is more modest. The most clinically relevant application today is using MSC-based approaches to treat fracture non-unions, cases where a broken bone fails to heal on its own. A 2025 systematic review and meta-analysis found that stem cell therapy reduced bone union time by a mean of about half a month (0.54 months) compared to controls, a statistically significant but modest effect. Allograft cellular bone matrix products (donor bone containing living cells and growth factors) are used in orthopedic surgery and regulated under FDA's human cells, tissues, and cellular products (HCT/P) framework. These are not the same as injecting stem cells into a joint and having them build new bone from scratch.
It is also worth knowing that stem cells exist in a broader growth context. Questions about where stem cells reside and how they proliferate before differentiating are closely related to understanding why certain environments support bone growth and others do not. Questions about where stem cells reside and how they proliferate before differentiating are closely related to understanding why certain environments support bone growth and others do not where do stem cells grow. Similarly, stem-cell-based cartilage repair, which shares many of the same MSC pathways and scaffold considerations, is a closely related area of research.
The real limits on stem-cell bone growth
Even setting aside everything about clinical translation, there are hard biological and engineering constraints on what stem-cell-derived bone can do today.
- Scale: Small defects and thin constructs are achievable. Large, complex bone segments (like rebuilding a load-bearing shaft) remain limited by vascularization and mechanical integration challenges.
- Integration: New bone must physically bond with host bone and be remodeled by the body over time. Poor integration leads to failure, even if the initial bone formation looks good.
- Differentiation reliability: In vitro markers like ALP activity and gene expression do not reliably predict whether a cell population will form bone in vivo. What works in a dish may not work in a body.
- Ectopic ossification risk: MSCs transplanted outside their normal anatomical location can still form bone, sometimes in places you do not want. Ectopic bone formation is documented in transplant studies and represents a real unintended consequence.
- Residual undifferentiated cells: With pluripotent stem cells (hESCs, iPSCs), any undifferentiated cells left in a transplanted population carry a risk of teratoma formation, a tumor that can contain mixed tissue types. This is taken seriously enough that early FDA-approved PSC trials require preclinical dose-escalation testing specifically to assess this risk.
- Durability: Long-term data on engineered bone constructs in humans remains limited. Most clinical evidence covers months, not decades.
Safety, regulation, and the ethics of stem-cell bone claims
Here is the part that matters most if you are looking at this from a practical health standpoint. As of today, there is no FDA-approved stem cell therapy for any orthopedic condition, including bone regeneration, osteoarthritis, fracture non-unions, or back pain. The FDA has explicitly stated this. The only FDA-approved stem cell products in the US are blood-forming stem cells from umbilical cord blood, used for hematopoietic conditions. The first FDA-approved MSC therapy, remestemcel-L (Ryoncil), was approved in December 2024 for a specific pediatric immune condition (steroid-refractory acute graft-versus-host disease), not for bone.
The FDA has taken enforcement action against clinics illegally marketing unapproved cellular products, including one case where a company was processing fat tissue into stromal vascular fraction (SVF) and administering it for a wide range of conditions under manufacturing conditions that put patients at risk. The FTC similarly requires that health claims be truthful, not misleading, and substantiated by evidence. Regulatory frameworks around bone-related products can also be complex: some allograft bone matrix products are regulated as HCT/Ps under Section 361 (minimal manipulation, homologous use), while others may be classified as devices or biologics depending on their composition and intended use. This matters because the level of clinical evidence required differs across those categories.
From an ethical standpoint, hESC-derived approaches involve human embryos, which remains a regulated and ethically debated area. iPSC approaches sidestep that issue by using reprogrammed adult cells, but they carry their own safety considerations around genomic stability and tumorigenicity.
What to actually do if you're exploring stem cells for bone repair
If you are a patient, a student researching this topic, or someone evaluating options after a difficult diagnosis, here is direct, practical guidance.
- Ask what type of stem cells are being used and where they come from. MSCs, hESCs, and iPSCs have different risk profiles, differentiation potentials, and regulatory statuses. A clinic that cannot clearly answer this question is a red flag.
- Ask what the regulatory status of the treatment is. Is it under an FDA-approved Investigational New Drug (IND) application? Is it part of a registered clinical trial (clinicaltrials.gov is the public database)? If neither, be very cautious.
- Ask for peer-reviewed evidence of in vivo bone formation, not just in vitro assays. As the research makes clear, ALP activity in a dish and gene expression readouts do not reliably predict what will happen in your body.
- Distinguish between allograft bone products (regulated HCT/Ps with real clinical track records) and experimental stem-cell injection therapies (often unregulated, unproven). These are not the same thing, even if both involve 'cells and bone repair.'
- Be skeptical of broad claims. Any clinic claiming stem cells can regenerate large bone segments, replace joints, or cure bone disease in a single injection, without referencing controlled clinical trial data, is overpromising.
- If you have a fracture non-union or bone defect, ask your orthopedic surgeon specifically about evidence-based options including bone grafting, bone morphogenetic protein therapies (BMP-2 is FDA-approved in specific spinal and fracture indications), and whether any clinical trials for MSC-based approaches are recruiting near you.
- For researchers and students: focus evaluation on studies that include in vivo histology with human-cell identification methods (like ALU hybridization), not just in vitro osteogenic marker assays. That is currently the most meaningful evidence of true bone formation from stem cells.
A quick comparison: stem cell types and bone-forming potential

| Stem Cell Type | Bone-Forming Potential | Evidence Level | Key Risks | Regulatory Status (US) |
|---|---|---|---|---|
| Mesenchymal Stem Cells (MSCs) | High, well-established osteogenic differentiation | Animal models, early clinical (fracture non-union) | Ectopic ossification, variable potency by donor/source | No approved orthopedic indication; some HCT/P products regulated under 361 |
| Human Embryonic Stem Cells (hESCs) | High, proven in vivo bone formation in animal models | Animal models, limited human data | Teratoma risk from residual undifferentiated cells, ethical/regulatory constraints | Experimental only; regulated under IND |
| Induced Pluripotent Stem Cells (iPSCs) | High potential, protocol-dependent in vivo bone formation | Animal models, early-stage human research | Teratoma risk, genomic instability, differentiation variability | Experimental only; regulated under IND |
| Allograft Cellular Bone Matrix (donor MSCs in bone matrix) | Moderate, supports bone repair via osteoconductivity and resident cells | Clinical use in orthopedic surgery | Donor screening dependent, handling protocol matters | Regulated as HCT/P under 21 CFR 1271 (Section 361 if qualifying) |
The bottom line is this: stem cells can grow bone, and the biology is real, well-characterized, and actively being translated into clinical tools. But that translation is partial, the conditions required are exacting, and the gap between a promising lab result and a safe, approved clinical treatment is wide. Knowing where a particular claim sits on that spectrum, lab study, animal model, clinical trial, or regulated approved therapy, is the most useful thing you can bring to any conversation about stem cells and bone.
FAQ
If stem cells can form bone in labs, why does it not reliably work when injected into a patient’s body?
Many injections deliver cells without the full package of instructions and support. In real bone formation, cells need a sustained osteogenic signaling environment, a place to attach and organize (scaffold or graft matrix), and enough oxygen and nutrients as the tissue grows. Without that, cells may show osteogenic markers in the short term but fail to build mineralized, structurally organized bone that can remodel.
How can you tell whether “bone formation” is real mineralized bone versus just early osteogenic activity?
True bone formation includes mineralized matrix and organized collagen structure, not only expression of osteogenic markers. In research, investigators confirm mineralization with stains that bind mineral or with tracer-based imaging, and they use histology to verify lamellar organization. In clinical contexts, imaging and clinical union timelines matter more than laboratory marker reports from cultures.
Why do some stem-cell therapies claim bone regeneration even when studies show only modest improvements?
A common issue is mixing endpoints. Some trials measure faster healing or improved radiographic union, not full “regrowth from scratch” at a large site. When effects are statistically significant but small, marketing can exaggerate what the therapy actually achieves, especially if the product is positioned as a broad solution for many orthopedic problems.
What are the most common red flags if a clinic is offering stem-cell bone therapy?
Be cautious if they promise guaranteed bone regrowth, claim approval for orthopedic use when none is FDA-approved for stem-cell orthopedic indications, or offer broad “one product for everything” protocols. Also watch for vague details about the cells’ source, processing method, dosing rationale, and how they address safety monitoring, because illegal or poorly controlled preparations have been an enforcement focus.
Does the source of the stem cells (MSCs vs iPSCs vs embryonic stem cells) change the risk profile?
Yes. iPSC-based approaches involve reprogramming adult cells, which raises specific concerns such as genomic stability and the theoretical risk of tumor formation if unwanted cell types are present. Embryonic stem cell approaches carry ethical and regulatory considerations. MSC approaches often emphasize implantation and differentiation performance within a graft-like environment. The key is that “bone potential” does not automatically equal “same safety.”
Can stem-cell-derived bone work for large defects like a whole vertebra, not just small injuries?
Scaling is limited by vascularization. Engineered bone larger than a few millimeters thick often develops a nutrient and oxygen gap in the interior before blood vessels can grow in, leading to nonviable or poorly mineralized tissue. This is why many lab constructs remain relatively small or rely on strategies to improve perfusion and integration.
If MSCs are the most common bone-forming stem cells, why aren’t simple MSC injections enough?
MSC injections often lack an engineered osteoconductive structure and an osteoinductive “training” regimen. Without a matrix to anchor cells and guide organization, and without controlled biochemical cues over time, many cells do not fully differentiate into mature osteoblasts that deposit mineralized, load-bearing bone.
What does it mean when a study shows osteogenic markers but no bone after transplantation?
It means differentiation signals in a dish did not translate into functional bone formation in vivo. Marker expression can reflect commitment steps, but mineral deposition, proper cell maturation, and tissue-level organization also require the right timing, factor concentrations, scaffold cues, and a vascular-capable environment.
Are all bone graft products the same if they contain “cellular components”?
No. Some products are regulated under categories like minimal manipulation and homologous use frameworks, while others may be treated as devices or biologics depending on composition and intended use. This affects what evidence is required and how claims can be made, so it matters whether the product is an approved cellular bone matrix product versus an unapproved, custom-prepared cell injection.
If stem cells reduce fracture non-union time by about half a month, how should a patient interpret that benefit?
That figure suggests a modest average improvement, not a dramatic effect for every patient. Real outcomes depend heavily on factors like the size and biology of the non-union, stability of fixation, time since injury, infection status, and the specific product protocol used. For decision-making, union rate and time-to-union in your specific clinical scenario usually matter more than broad promises.
What questions should I ask a clinician if I’m considering a stem-cell-related bone option?
Ask whether the therapy is FDA-approved for your indication, what specific product it uses and under what regulatory category, the cell source and processing steps, the evidence level for your exact condition type, how they manage infection and fixation requirements, and what imaging timeline they use to judge union. Also ask what alternatives exist and what risks are emphasized for that product.
Citations
Osteoblast-related lineage and “bone-forming” markers used in practice/literature include proteins such as type I collagen, bone sialoprotein (BSP), osteocalcin (OC), and osteopontin (OPN), along with mineralization labels (e.g., alizarin complexone/calcein/demeclocycline) to identify mineralized matrix.
https://www.ncbi.nlm.nih.gov/books/NBK557792/
In osteogenic differentiation pathways, early osteoblast-associated markers include ALP, BSP, and type I collagen (Col1a1), while osteocalcin (OCN) appears later concomitantly with mineralization; functional “true bone formation” requires more than just osteogenic marker expression.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3392028/
A defining biological meaning of “stem-cell-derived bone” in vivo is histologically proven bone of human origin with highly mineralized matrix (e.g., fluorescence consistent with mineralization) and lamellar collagen organization observable weeks after transplantation (reported at 8, 12, and 16 weeks).
https://pmc.ncbi.nlm.nih.gov/articles/PMC3128756/
For iPSC-derived bone, the “gold standard” evidence cited is actual bone formation in vivo (not just qPCR/i in vitro readouts), with the study noting that qPCR-based assays at the end of differentiation did not reliably predict bone formation in vivo.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4073820/
Runx2 is described as directing multipotent mesenchymal cells into the osteoblast-lineage, while Sp7 (Osterix) is required downstream; mouse knockouts of Runx2 and Sp7 lack osteoblasts and bone formation, supporting causality of these lineage transcription factors.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11432631/
A commonly used “osteogenic differentiation” claim is anchored on two master transcription factors—Runx2 and Sp7/Osterix—whose expression/function control downstream osteoblast-phenotype genes.
https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2020.611197/full
In vitro osteogenic differentiation of hESCs was reported to upregulate osteoblastic marker genes including Runx2, osterix, bone sialoprotein (BSP), and osteocalcin, accompanied by formation of mineralized bone matrix.
https://pubmed.ncbi.nlm.nih.gov/17233553/
The hESC study demonstrates that hESC-derived differentiated cell regimes produced histologically proven human-origin bone in vivo, with observation timepoints at 8, 12, and 16 weeks post-transplant.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3128756/
Bone formation in vivo was achieved using specific iPSC differentiation media schedules (including programs involving BMP4 and bFGF, followed by Dex+AscP in the reported approach), reinforcing that differentiation regimen affects whether “true bone” forms in vivo.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4073820/
To confirm bone formation attributable to human cells, the study used human-specific ALU repetitive DNA in situ hybridization to detect human cells in transplanted tissue.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4073820/
A typical in vitro osteogenic differentiation readout includes ALP activity and mineralization assays; the study reports higher alkaline phosphatase activity on days 7 and 14 on MgO/PCL nanofibrous scaffolds versus pure PCL.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9012933/
Wnt/β-catenin signaling is described as acting later in the differentiation pathway to osterix+ osteoblast precursors and then toward bone-secreting osteoblasts, while Ihh acts earlier as a switch initiating bone formation programs via Runx2+ osteoblast precursors.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3392028/
The review states that alkaline phosphatase (ALP), BSP, and collagen type 1 alpha 1 (Col1a1) are early osteoblast differentiation markers, and osteocalcin (OCN) appears late during the mineralization-associated stage.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3392028/
Experimental literature links BMP2 signaling to Osterix (Sp7) regulation via Msx2 and Runx2 pathways during osteoblast differentiation, supporting BMP2 as an osteoinductive cue.
https://pubmed.ncbi.nlm.nih.gov/18703512/
Scaffold pore-size relates to tissue outcome: one review reports that smaller pore sizes (<100 μm) can be associated with formation of non-mineralized osteoid or fibrous tissue, highlighting that scaffold physical parameters can determine whether bone mineralizes.
https://www.sciencedirect.com/science/article/pii/S2468217920300071
A review reports macroporosity (pores >50 μm) as important for cell colonization and growth of vascular and bone tissue, linking pore architecture to vascularization and osteogenesis.
https://www.sciencedirect.com/science/article/pii/S0928493115306937
A scaffold-design review emphasizes that porosity, pore size, and pore shape should be optimized to support cell viability and proliferation as part of bone tissue engineering protocol design.
https://link.springer.com/article/10.1007/s12541-022-00755-7
A recent scaffold-review frames successful osseous tissue regeneration around osteoconductivity (porous support), osteoinductivity (growth factors inducing MSC differentiation into osteoblasts), osteogenicity (osteoblast activity for mineral deposition), and osteointegration (bonding with host).
https://www.mdpi.com/2306-5354/13/5/514
FDA states that some products containing human demineralized bone matrix (DBM) combined with other components intended to change handling may not qualify to be regulated solely under Section 361; FDA also describes four criteria used for HCT/P (361) regulation categorization.
https://www.fda.gov/combination-products/jurisdictional-updates/jurisdictional-update-human-demineralized-bone-matrix
FDA guidance notes that minimally manipulated demineralized bone is regulated as an HCT/P under Section 361 (21 CFR 1271.10), illustrating that many “bone graft” products may not be regulated as stem-cell therapies even if they contain cellular components or are marketed for bone repair.
https://www.fda.gov/medical-devices/guidance-documents-medical-devices-and-radiation-emitting-products/dental-bone-grafting-material-devices-class-ii-special-controls-guidance-industry-and-fda-staff
FDA states that regenerative medicine therapies have not been approved for treatment of any orthopedic condition (including osteoarthritis, tendonitis, disc disease, tennis elbow, back pain, hip/knee/shoulder pain).
https://www.fda.gov/vaccines-blood-biologics/consumers-biologics/important-patient-and-consumer-information-about-regenerative-medicine-therapies
FDA indicates RYONCIL (remestemcel-L-rknd) is approved for steroid-refractory acute graft-versus-host disease (SR-aGvHD) in pediatric patients 2 months of age and older—demonstrating that FDA-approved MSC therapy exists but not for orthopedic bone regeneration indications.
https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/ryoncil
FDA approval date specified: December 18, 2024 for remestemcel-L-rknd (Ryoncil) in the pediatric SR-aGvHD indication.
https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-remestemcel-l-rknd-steroid-refractory-acute-graft-versus-host-disease-pediatric
NuVasive states Osteocel products are “allograft cellular bone matrix,” describes donor qualification and multistep screening per FDA/AATB regulations, and claims large treated-patient numbers and peer-reviewed publications (company marketing claims).
https://www.nuvasive.com/procedures/featured-offerings/osteocel/
A 2025 systematic review/meta-analysis of controlled studies reported stem cell therapy reduced bone union time by a mean difference of 0.54 months (95% CI −0.75 to −0.33), while noting heterogeneity across studies.
https://bmcmusculoskeletdisord.biomedcentral.com/articles/10.1186/s12891-025-08365-w
The review explains pluripotent stem cell (PSC) tumorigenicity risks include malignant transformation and teratoma formation from residual undifferentiated cells, describing teratoma formation as a “gold standard” for pluripotency assessment and highlighting preclinical dose-escalation testing in early FDA-approved PSC trials.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3967018/
MSC studies show ectopic bone formation capability after ectopic transplantation into immunodeficient mice when using HA/TCP particles as a vehicle—illustrating both the potential and a failure-mode risk (unwanted ectopic ossification).
https://pmc.ncbi.nlm.nih.gov/articles/PMC4618923/
The iPSC bone study warns against relying solely on in vitro gene expression/qPCR to predict in vivo bone formation, implying a failure mode where differentiation “markers” may not translate to safe/consistent in-body outcomes.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4073820/
FDA provides regulatory resources for cellular/gene therapy products, including related guidance links and policy/enforcement context relevant to stem cell clinics’ oversight and compliance.
https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/resources-related-regenerative-medicine-therapies
FDA press announcement (Nov 13, 2018) describes enforcement against a company illegally marketing an unapproved cellular product and cites manufacturing conditions that put patients at risk, including processing adipose tissue into SVF for multiple routes/indications.
https://www.fda.gov/news-events/press-announcements/fda-warns-stemgenex-biologic-laboratories-llc-illegally-marketing-unapproved-cellular-product
FDA states many regenerative medicine products are illegally marketed as safe/effective for wide disease ranges even though they haven’t been adequately studied under an IND to demonstrate safety/effectiveness.
https://www.fda.gov/vaccines-blood-biologics/consumers-biologics/important-patient-and-consumer-information-about-regenerative-medicine-therapies
FDA’s jurisdictional discussion underscores due diligence: bone matrix products may be regulated as devices or under HCT/P frameworks rather than as “stem cell therapies,” affecting what clinical evidence and regulatory approvals you should expect.
https://www.fda.gov/combination-products/jurisdictional-updates/jurisdictional-update-human-demineralized-bone-matrix
FDA indicates the only FDA-approved “stem cell products” for use in the US are blood-forming stem cells from umbilical cord blood (hematopoietic progenitor cells), not orthopedic stem-cell bone regeneration therapies.
https://www.fda.gov/vaccines-blood-biologics/consumers-biologics/important-patient-and-consumer-information-about-regenerative-medicine-therapies
FTC guidance emphasizes that health-related labeling/advertising must be truthful, not misleading, and substantiated—relevant to evaluating marketing claims made by stem-cell clinics.
https://www.ftc.gov/business-guidance/resources/health-products-compliance-guidance
TGA guidance provides an example compliance framework for advertising of stem-cell/HCT products, reinforcing that regulators treat stem-cell claims as subject to advertising restrictions and enforcement for non-compliance.
https://www.tga.gov.au/resources/guidance/advertising-stem-cell-and-other-human-cell-or-tissue-hct-products
Where Do Stem Cells Grow: Body Niches and Lab Culture
Discover body niches and lab conditions that let stem cells self-renew and avoid differentiation.