Cells do two distinct things when we say they "grow and reproduce": they physically get bigger, and then they split into two. Both steps are real, separate processes, and understanding each one makes the whole picture click. The short answer is that a cell grows by taking in nutrients and building new proteins, lipids, and organelles; it reproduces by copying its DNA and then physically dividing into two daughter cells through a sequence called the cell cycle. What prevents this from happening forever? Checkpoints, resource limits, and built-in molecular brakes that halt division when something goes wrong. Let's walk through all of it.
How Do Cells Grow and Reproduce: Cell Cycle Explained
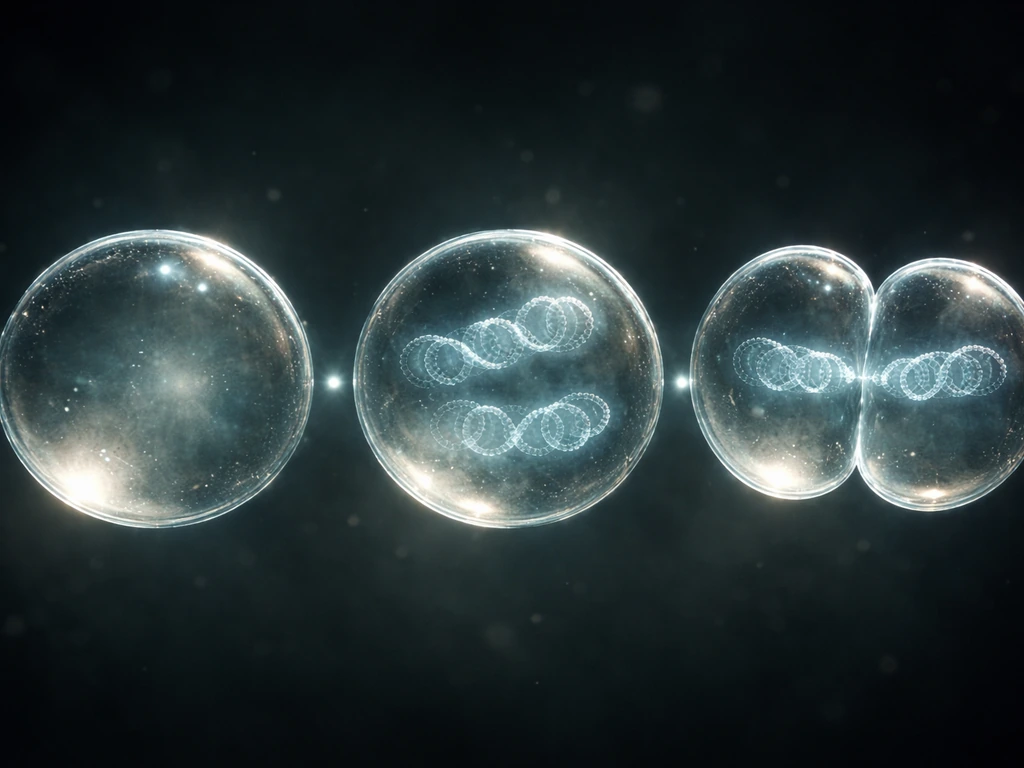
Cell growth vs. cell division: what "grow and reproduce" actually means

Growth and reproduction are not the same event, even though they're tightly linked. Growth means a cell increases its mass and volume: it synthesizes proteins, builds new membranes, duplicates organelles, and accumulates the molecular machinery it will need to function as two cells instead of one. Reproduction means the cell copies its genetic information and then physically splits, producing two daughter cells that each carry a complete set of DNA.
Think of it like baking a loaf of bread and then cutting it in half. The "baking" part is growth. The cutting is division. If you skip the baking and just cut, you get two undersized pieces. Cells have the same constraint: a daughter cell that didn't grow properly before division will be too small and resource-poor to function well. That's why growth and division are so tightly coordinated, and why the cell cycle has dedicated checkpoints to make sure growth is complete before division begins. Whether we're talking about a bacterium dividing on a petri dish or a human skin cell replacing itself, the core logic is the same.
It's also worth noting that not every cell reproduces at the same rate or even at all. The ability to grow and reproduce is not universal across all cell types in your body: neurons rarely divide after development, while intestinal lining cells replace themselves every few days. The type of cell, its environment, and its developmental history all determine whether it will enter the cell cycle.
The cell cycle: G1, S, G2, and M in plain terms
The cell cycle is the organized sequence of events a cell goes through from the moment it's born (as a daughter cell) to the moment it divides again. There are four main phases, and each one has a job.
- G1 (Gap 1): The cell grows in size, produces proteins and organelles, and gets assessed for readiness. This is the longest phase for most cell types. A key regulatory event called the restriction point (controlled by cyclin D/CDK4/6 activity and the Rb protein) determines whether the cell commits to dividing or holds off.
- S (Synthesis): The cell copies its entire DNA. Every chromosome gets duplicated so that the eventual daughter cells each receive a full genome.
- G2 (Gap 2): The cell grows more, checks that DNA replication was completed accurately, and assembles the physical machinery it needs for division.
- M (Mitotic phase): The cell actually divides — first the nucleus splits (mitosis), then the cell body splits (cytokinesis), producing two daughter cells.
Transitions between these phases are controlled by cyclin-CDK complexes: proteins whose concentration rises and falls at specific points in the cycle. For example, in budding yeast, S-phase cyclins like Clb5 and Clb6 form complexes with CDKs that directly trigger the start of DNA replication as the cell enters S phase. Cell cycle checkpoints sit at the G1/S boundary, the G2/M boundary, and inside M phase itself (the spindle checkpoint), verifying that each previous phase was completed correctly before the next one begins.
How cells copy their DNA before dividing

DNA replication is the centerpiece of S phase, and it's more complicated than "unzip and copy." Here's what actually happens at the molecular level.
Replication starts at specific sites on the chromosome called origins of replication. Prokaryotes like E. coli typically have a single origin and replicate from there in both directions. Eukaryotes (including human cells) fire thousands of origins simultaneously, which is the only way to replicate billions of base pairs in a reasonable amount of time. At each origin, a replisome complex unwinds the double helix and sets up a replication fork that moves outward in both directions.
At each fork, the two DNA strands are copied differently. The leading strand is synthesized continuously in the direction the fork is moving. The lagging strand runs in the opposite direction relative to the fork's movement, so it has to be copied in short, discontinuous segments called Okazaki fragments. An enzyme called primase lays down a short RNA primer, then DNA polymerase extends it into a new DNA fragment. Those RNA primers are later removed, the gaps are filled with DNA, and DNA ligase seals the fragments together. Replicating the human genome involves processing roughly 30 million Okazaki fragments per cell division cycle, which gives you a sense of the scale of this operation.
Once replication is complete, the cell has two full copies of its genome, each consisting of two sister chromatids held together. Those copies then need to be separated accurately, which is mitosis's job. Before we get there, though, it helps to understand what the cell is actually doing to grow during G1 and G2.
Mitosis and cytokinesis: one cell becomes two
Mitosis is nuclear division. Its goal is to take the two copied sets of chromosomes and sort them into two separate nuclei so that each daughter cell gets exactly one complete genome. It unfolds across four main stages.
- Prophase: Chromosomes condense from their loose, extended form into compact, visible structures. In the cytoplasm, microtubules begin organizing into the mitotic spindle.
- Prometaphase and metaphase: The nuclear envelope breaks down. Spindle microtubules attach to chromosomes at specialized protein structures called kinetochores. The chromosomes are then pulled and pushed until they line up at the cell's middle, the metaphase plate. The dynamic tension in the spindle at this point is what sets up the machinery for anaphase.
- Anaphase: The spindle pulls sister chromatids apart to opposite ends of the cell. Each chromatid is now its own individual chromosome, and each pole of the cell has a full set.
- Telophase: A new nuclear envelope forms around each set of chromosomes. The chromosomes begin to decondense. The cell now has two nuclei.
Cytokinesis is the physical splitting of the cell body that follows (or overlaps with) telophase. In animal cells, a contractile ring made of actin and myosin II filaments assembles around the cell's middle and constricts like a purse-string, pinching the cell into two. The timing and regulation of when this ring forms and how it disassembles are tied directly to the mitotic transitions happening in the nucleus, so the whole process is coordinated rather than sequential. In plant cells, cytokinesis works differently: instead of a contractile ring, a new cell wall is built from the inside out along the division plane.
After cytokinesis, you have two genetically identical daughter cells, each in their own G1 phase, ready to grow and potentially repeat the cycle.
What cells actually need to grow: nutrients, energy, and biosynthesis

Cell growth isn't passive. A cell doubling its mass means it has to build an enormous amount of new molecular machinery: proteins, lipids for new membranes, ribosomes, and copies of organelles. All of that requires raw materials and energy.
The central coordinator of cell growth in eukaryotes is a protein kinase called mTOR (mechanistic target of rapamycin). mTOR acts as a nutrient sensor: when amino acids, glucose, and growth factors are available, mTOR is active and it drives protein synthesis by activating S6K1 and releasing 4E-BP1's inhibition of eIF4E, a key translation initiation factor. In plain terms: mTOR reads the environment, and when conditions look good, it tells the cell to ramp up protein production and grow. It also links cell growth directly to cell cycle progression, so a cell that isn't growing well won't rush into division.
If you've ever wondered what human cells need to grow and reproduce at a practical level, the list comes down to: sufficient amino acids for protein synthesis, glucose (or other carbon sources) for energy via ATP, lipid precursors for membrane biosynthesis, nucleotides for DNA and RNA, and appropriate signaling from growth factors that tell the cell the organism actually needs more cells right now.
There's also a size-control problem. Cells can't just grow indefinitely and then divide whenever. They appear to use a combination of timer-like mechanisms and size-sensing (the exact mechanisms are still being studied) to ensure that division happens at an appropriate size. If a cell is too small when it enters G1, it delays the restriction point and stays in G1 longer, accumulating more mass before committing to replication.
What stops cells from reproducing endlessly
This is arguably the most important part of understanding how cells grow and reproduce, because the consequences of unchecked division are severe. Cancer is essentially what happens when these stopping mechanisms fail. There are several layers of control.
Cell cycle checkpoints

Checkpoints are molecular surveillance systems embedded at critical transitions in the cell cycle. The G1/S checkpoint evaluates whether DNA is intact and whether the cell has grown enough before committing to replication. The G2/M checkpoint verifies that DNA replication was completed and that there's no significant DNA damage before mitosis begins. The spindle assembly checkpoint (inside M phase) confirms that every chromosome is properly attached to the spindle before anaphase is allowed to proceed.
When DNA damage is detected, checkpoint kinases spring into action. ATR kinase (activated by replication stress and single-strand DNA) and ATM kinase (activated by double-strand breaks) signal downstream to checkpoint kinase 1 (Chk1) and Chk2. Chk1, for example, enforces the G2/M checkpoint by inhibiting the phosphatase Cdc25, which in turn keeps CDK1 in an inactive (phosphorylated) state, blocking mitosis entry. This Chk1/Wee1/Cdc25/CDK1 axis is one of the best-studied braking mechanisms in cell biology, and mutations that disrupt it are found in many cancers.
Understanding how cells grow and reproduce to maintain homeostasis is really about understanding how these checkpoints keep division proportional to the organism's needs, rather than letting any one cell line run wild.
Senescence: the permanent brake
When a cell accumulates too much damage, or when it has divided a certain number of times (as telomeres shorten), it can enter a state called senescence: a stable, permanent cell-cycle arrest. Senescent cells don't die right away, but they stop dividing. The key molecular actors here are the p53/p21 pathway and the p16INK4A pathway, both of which inhibit CDK4/6 activity and prevent the phosphorylation of Rb that would otherwise allow S-phase entry. Senescence can occur at G1 or G2, and it's a tumor-suppressor mechanism: the cell essentially takes itself out of the game rather than risk passing on damaged DNA.
Resource limits and contact inhibition
Even in the absence of DNA damage, cells stop proliferating when resources run low or when they become too crowded. In fibroblasts (and many other cell types), high cell density triggers proliferation arrest partly through upregulation of CDK inhibitors like p27Kip1. This contact inhibition is one reason normal cells in a dish stop growing once they form a monolayer, while cancer cells (which often lack this response) pile up on top of each other. From an organism's perspective, this is how tissues maintain their correct size and architecture during development.
Much of what cells need before they can grow and reproduce is about satisfying these checkpoint gatekeepers: enough nutrients, intact DNA, the right signals from neighboring cells and from the organism-level hormonal environment.
Growth and division across different organisms and contexts
The cell cycle logic above applies broadly, but the details vary a lot depending on the organism and the type of cell.
Bacteria: faster and simpler
E. coli doesn't have a nucleus or membrane-bound organelles, so its division process is streamlined. It replicates from a single chromosomal origin and divides by binary fission rather than mitosis. The timing is described using C and D periods: the C period is the time taken for the chromosome to fully replicate, and the D period is the gap between replication completion and the actual division event. Interestingly, the positioning of the division ring (the Z-ring, made of FtsZ protein rather than actin) is coordinated with chromosome replication: Noc proteins bind specific chromosomal regions and physically delay Z-ring assembly until the chromosome is clear of the division site. Even in a cell without a nucleus, division and replication have to be coordinated.
If you're curious about a eukaryotic microorganism that bridges the gap between bacteria-like simplicity and full eukaryotic complexity, how yeast cells grow and reproduce is a great case study, yeast have been the workhorse model for discovering most of what we know about eukaryotic cell cycle checkpoints and cyclin-CDK regulation.
Multicellular organisms: division in service of the whole
In a multicellular animal or plant, cell division isn't just about making more cells for its own sake. It's coordinated at the organismal level through hormones, growth factors, and cell-to-cell signaling. During development, different tissues divide at different rates and stop at different times. Stem cells are a particularly interesting case: they maintain the ability to divide and differentiate long after most other cells have stopped. What stem cells need to grow and divide is a more nuanced question than what a generic somatic cell needs, because stem cells also have to balance self-renewal (making more stem cells) against differentiation (making specialized cells).
There's also the question of what organelles do during cell growth. As a cell grows before division, it duplicates not just its DNA but also its mitochondria and (in plant cells) chloroplasts. These organelles have their own DNA and replicate somewhat independently. Organelles that grow and divide to reproduce themselves follow a similar logic to the overall cell cycle: they increase in size and then pinch in two, ensuring daughter cells inherit a working set of energy-producing machinery.
Cytokinesis across species
Even within eukaryotes, cytokinesis looks different. Animal cells use an actin-myosin contractile ring. Plant cells build a new wall down the middle. Budding yeast (Saccharomyces cerevisiae) bud off a daughter cell from one side of the mother cell and then divide there, while fission yeast (Schizosaccharomyces pombe) divide symmetrically. Both yeasts use contractile ring mechanisms, but the timing and positioning rules differ between them. The diversity is a reminder that the principle (split one cell into two) is conserved, but the molecular implementation is flexible.
Putting it all together: the sequence you actually need to know
Here's the practical summary of how a cell grows and reproduces, in order:
- G1: The newborn daughter cell grows, synthesizes proteins and organelles, and responds to nutrient availability via mTOR. Cyclin D/CDK4/6 activity gradually overcomes Rb inhibition. At the restriction point, the cell commits to dividing.
- S phase: DNA replication fires from thousands of origins. Leading strands are copied continuously; lagging strands are built from millions of Okazaki fragments, all of which are processed and ligated. The cell ends S phase with two full copies of its genome.
- G2: More growth and final preparation. The G2/M checkpoint verifies replication was error-free. If DNA damage is detected, Chk1 and related kinases freeze CDK1 activity and halt the cycle.
- Prophase to telophase (mitosis): Chromosomes condense, the spindle forms, kinetochores attach, chromosomes line up, sister chromatids separate, and two new nuclei form.
- Cytokinesis: The contractile ring (in animal cells) or new cell wall (in plant cells) physically splits the cytoplasm. Two daughter cells are born, each in G1, ready to grow.
A quick comparison: cell division in prokaryotes vs. eukaryotes
| Feature | Prokaryotes (e.g., E. coli) | Eukaryotes (e.g., human cells) |
|---|---|---|
| DNA structure | Single circular chromosome, no nucleus | Multiple linear chromosomes, enclosed in nucleus |
| Origins of replication | Typically one origin | Thousands of origins fired simultaneously |
| Division mechanism | Binary fission via FtsZ Z-ring | Mitosis + cytokinesis (actin-myosin ring or cell plate) |
| Cell cycle checkpoints | Less formalized; timing linked to C and D periods | Defined G1/S, G2/M, and spindle checkpoints |
| Duration | Can be as fast as 20 minutes under ideal conditions | Hours to days depending on cell type |
| Organelle duplication | Not applicable (no membrane-bound organelles) | Mitochondria, chloroplasts replicate independently |
The cell cycle, replication, mitosis, and cytokinesis are among the most fundamental processes in all of biology. Once you understand that growth and division are two separate but coordinated steps, and that both are gated by nutrient availability, checkpoint integrity, and organism-level signals, the whole picture of how cells grow and reproduce becomes much clearer. The reason cells don't divide forever isn't a failure of design; it's the design. Checkpoints, senescence, and contact inhibition are the features, not the bugs.
FAQ
If growth and division are separate, how does a cell prevent starting DNA replication before it has built enough cellular components?
Cells use checkpoint delays to couple growth to readiness. If growth is lagging, the cell tends to spend longer in G1 before it passes the restriction point, so it delays committing to replication rather than starting S phase early with an undersupplied biosynthetic program.
Do cell cycle checkpoints only respond to DNA breaks, or do they also respond to replication problems?
Checkpoint proteins do not act only by sensing “DNA damage.” For instance, during replication stress ATR signaling responds to problems like stalled or slowed replication forks, so the cell can pause before mitosis even when the DNA is not visibly broken.
If senescence stops division, does it always require DNA damage, and does it mean the cell will die?
No. Many cells can enter senescence or a growth arrest even without DNA lesions, for example when stress pathways are activated or when resources are limited. Also, senescence is different from apoptosis, because the cell remains alive but permanently stops cycling.
How does the cell make sure cytokinesis does not separate cells with the wrong chromosomes?
Cytokinesis must deliver two viable cell halves, so cells often couple it to nuclear events. If chromosome segregation is incorrect or the spindle is not properly attached, spindle checkpoint signaling halts progression to anaphase, which helps avoid producing daughter cells with mismatched chromosome sets.
Why can cells fail to grow and reproduce even if their DNA is intact and the checkpoints are working?
Eukaryotic growth relies on sustained biosynthesis, especially translation capacity, not just DNA copying. In nutrient-poor conditions, mTOR activity drops and protein synthesis slows, which makes it harder to reach the size and molecular prerequisites needed to pass into replication.
What specifically causes contact inhibition when cells get too crowded, is it only physical space limits?
Cell density effects are not purely mechanical. High density can change signaling, upregulate cyclin-dependent kinase inhibitors like p27Kip1, and reduce proliferative cues from the environment, leading to contact inhibition-like arrest in many normal cell types.
Does telomere shortening alone determine how many times a cell can divide?
Telomere shortening is one trigger that pushes cells toward senescence after a limited number of divisions, but it is not the only route to permanent arrest. Cells can also senesce due to other stress inputs, for example strong oncogenic signaling or sustained culture stress.
How is division coordination different between bacteria (binary fission) and eukaryotes (mitosis)?
Prokaryotes and eukaryotes both coordinate replication with division, but they do not share the same structures. Bacteria use Z-ring formation and division-site positioning controls (like Noc protein-mediated delay), while eukaryotes use mitosis, spindle attachment, and different checkpoint logic.
Do mitochondria and other organelles always get split equally during cell division?
Organelles follow a “growth then partition” principle, but organelle inheritance can be asymmetric. For example, mitochondria can be inherited based on distribution and fission-fusion dynamics, so daughter cells may not receive identical organelle counts even when the overall process is coordinated.
What happens if a cell is too small when it reaches the G1 checkpoint?
A cell that is too small at the G1 restriction point tends to delay entry into S phase, which increases the time available to accumulate mass and replenish components. This helps ensure that when division happens, daughter cells have sufficient resources to re-establish their own growth programs.
Before Human Cells Can Grow and Reproduce, They Need What
Cells need nutrients, energy, water and ions, proper pH O2, signals, attachment, and a working genome to divide.