Mycelium grows by extending tiny thread-like structures called hyphae, one microscopic tip at a time. Each hyphal tip pushes forward, branches repeatedly, and the resulting network colonizes whatever organic material is available. That is the short answer. But understanding the mechanism, the conditions, and the constraints behind that process is what actually helps you grow it successfully, diagnose problems, or just make sense of the white fuzzy stuff taking over your compost pile. Let's walk through all of it.
How Does Mycelium Grow: Growth Steps, Needs, and Troubleshooting

What mycelium actually is and how it builds itself
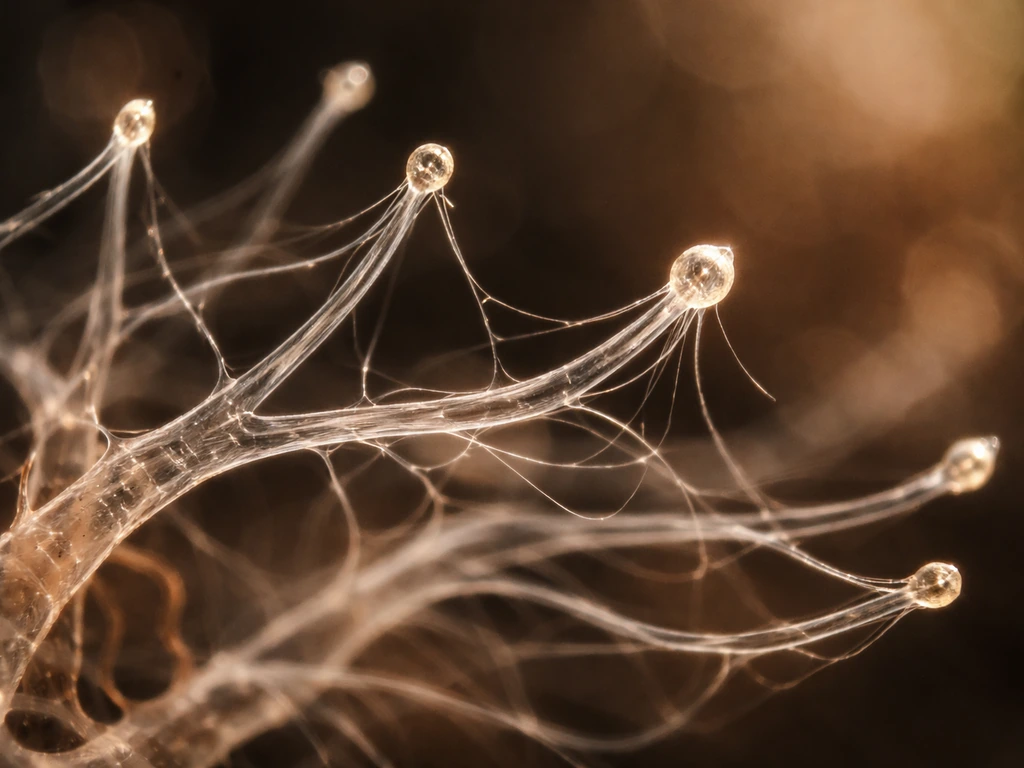
Mycelium is the collective term for a network of hyphae, and hyphae are the main mode of vegetative growth in most fungi. Think of a single hypha as a living tube. It does not grow uniformly along its length the way a plant stem might. Instead, all growth is concentrated at the very tip, a process biologists call tip growth or polarized growth.
At the hyphal apex, there is a specialized structure called the Spitzenkörper, a dense cluster of secretory vesicles sitting right at the growing tip. Its position directly controls the direction and rate of growth. The Spitzenkörper works like a cellular delivery hub: it continuously supplies vesicles carrying cell-wall-building enzymes and new membrane material to the apex. As those vesicles fuse with the tip, the wall extends forward, and the hypha literally pushes itself deeper into the substrate. Enzyme release happens right at the tip too, beginning the chemical breakdown of whatever organic material lies ahead.
This whole process depends on three things working together: cell-wall synthesis at the apex, the internal movement of vesicles toward the tip, and turgor pressure, the hydraulic pressure inside the hypha that physically drives the tip forward. Change any one of those, and growth slows or stops. If you want a deeper look at how individual hyphae behave at the cellular level, the article on how do hyphae grow covers the cellular mechanics in more detail.
Branching: how one thread becomes a network
As a hypha extends, it periodically initiates new side branches. Branching in fungi is essentially equivalent to cell division in other organisms: it is how the organism multiplies its growing points and expands its reach. Each branch is a new tip that can extend independently, and those branches branch again. A spore germinates, throws out a single hypha, and within days that single structure becomes a radiating web covering the available surface. The complexity of the mycelial network is entirely built from this repeated tip-extension-plus-branching loop.
When mycelium encounters a solid substrate like wood, it does not just grow along the surface. Hyphae penetrate between cells through natural openings called pits and, where needed, bore directly through cell walls. There is a real physical constraint here: no fungus can penetrate micropores smaller than about 0.2 micrometers, meaning the porosity and cell structure of a substrate acts as a filter that determines how quickly and thoroughly mycelium can colonize it. Dense, refractory woods take longer precisely because the pit membranes and cell walls resist penetration.
Where mycelium grows in nature

The honest answer is: wherever there is organic material, moisture, and oxygen. In forests, mycelium is found in soil, underneath leaf litter, inside decaying wood, and woven through compost. Leaf litter is one of the richest natural substrates because it concentrates organic carbon, nitrogen, and phosphorus exactly where fungal decomposers need them. Flip over a log after rain and you will almost always find pale white threads running through the rotting wood underneath.
Mycorrhizal species grow in even more specific locations. Ectomycorrhizal fungi, for example, form what is called an extramatrical mycelium that extends centimeters outward from plant roots through the surrounding soil and leaf litter. That web of hyphae is not randomly wandering. It is actively exploring for phosphorus and water, then shuttling those resources back to the host root through the Hartig net, the structured contact zone where the fungus and root exchange nutrients. This is one of the most elegant growth systems in biology: the plant feeds the fungus carbon, and the fungus dramatically extends the plant's effective root zone.
Indoors, the same principle applies but the substrates change. Fungi can colonize OSB, the paper backing on gypsum drywall, cellulose ceiling tiles, carpet, plywood, and paneling, anywhere moisture is present and cellulose or other organic compounds are available. The EPA is direct about this: moisture intrusion is the trigger. Fix the moisture source and you address the growth condition. Ignore it and the mycelium will keep colonizing as long as substrate is available.
For cultivators growing edible mushrooms, the most productive substrates are lignocellulosic materials: hardwood sawdust, straw, wheat bran supplements, and agricultural waste products. Research on oyster mushrooms (Pleurotus species) shows that substrate composition based on lignocellulosic waste directly influences how well mycelium colonizes and how much it ultimately produces. The fungus matches its digestive enzyme output to what the substrate offers.
What the environment needs to look like for growth to happen
Five factors control whether mycelium grows, and if any one of them falls outside the acceptable range, growth either slows dramatically or stops entirely. Here is how each one works in practice.
Water availability

Water is the single most important factor. Fungi need unbound water, measured as water activity (aw) rather than total moisture content. Most molds stop growing below about aw 0.8, though the exact threshold varies by species. In practical cultivation, relative humidity in the growing environment is the lever you control. Oyster mushroom cultivation typically targets 80 to 85% relative humidity during colonization, maintained by misting the growing space multiple times per day. Too dry and hyphal tips cannot maintain turgor pressure. Too wet and you create a different problem: waterlogged substrate reduces porosity and cuts off oxygen supply.
Oxygen and CO2 balance
Fungi are aerobic organisms. They need oxygen and produce CO2 as a metabolic byproduct. In a sealed container or a dense, wet substrate, CO2 builds up and growth slows. Good air exchange is not optional. During colonization, the goal is providing enough fresh air that CO2 does not accumulate to inhibitory levels. Research confirms that elevated CO2 concentration directly reduces hyphal extension rates, and this is one reason why tight lids or bags without filter patches consistently produce stalled or patchy growth.
Temperature
Each fungal species has a preferred temperature window for mycelial growth that is often different from its preferred fruiting temperature. Most commonly cultivated species colonize fastest somewhere between 21 and 27°C (roughly 70 to 80°F), though research shows hyphal extension rates are measurably affected at 30, 37, and 42°C with the differences becoming more dramatic at extreme values. Being even slightly outside the optimal range consistently slows colonization. Cold slows enzyme activity. Heat denatures proteins, damages the mycelium, and opens the door to competing organisms.
pH
Most saprotrophic fungi prefer mildly acidic to near-neutral conditions, roughly pH 5 to 7. pH directly affects the enzymes responsible for cell-wall synthesis at the hyphal tip, so a strongly alkaline or strongly acidic substrate can effectively shut down tip growth even when every other condition is perfect. Research tracking hyphal extension rates across pH levels confirms that the relationship is not subtle: wrong pH, dramatically reduced or zero extension.
Nutrients
Mycelium needs a carbon source for energy and structure, plus nitrogen for protein synthesis, and trace minerals. In nature, decaying wood and leaf litter supply these in varying ratios. In cultivation, substrate recipes are essentially nutrient management: you adjust the carbon-to-nitrogen ratio to match what the target species needs while avoiding excess nitrogen that tends to invite bacterial contamination. The mycelium secretes digestive enzymes ahead of its advancing tips, breaking down complex polymers into soluble nutrients it can absorb. It is digestion-first, absorption-second, and the tip is leading the charge.
Network growth vs. fruiting: they are not the same thing
A lot of confusion comes from conflating vegetative mycelial growth with mushroom fruiting. They are two distinct phases with different environmental triggers. Vegetative growth is the colonization phase: hyphae expanding through substrate, building the network, digesting organic material. Fruiting is the reproductive phase: the mycelium consolidates resources and constructs a mushroom (or other fruiting body) to release spores.
The switch from colonization to fruiting is not automatic. It requires a change in conditions. For many cultivated species, fruiting is triggered by a temperature drop (sometimes called a cold shock), increased fresh air exchange, higher humidity (often up to 90 to 95%), and in some species, the presence of light. Button mushrooms, for example, are typically moved to cooler fruiting temperatures of 16 to 18°C only after full colonization is confirmed. Light plays a complex role too: it can induce the formation of primordia (the early pin stage) in some species, while in others it can actually reduce yield. The critical practical point is this: if you have a fully colonized substrate that is not fruiting, look at your environmental conditions first, not the mycelium itself.
This distinction matters on the biology side too. Moisture that supports colonization may actually work against fruiting if it reduces substrate porosity and limits oxygen transfer to developing fruit bodies. The same substrate needs different management at different stages.
How fast does mycelium grow and what puts the brakes on it
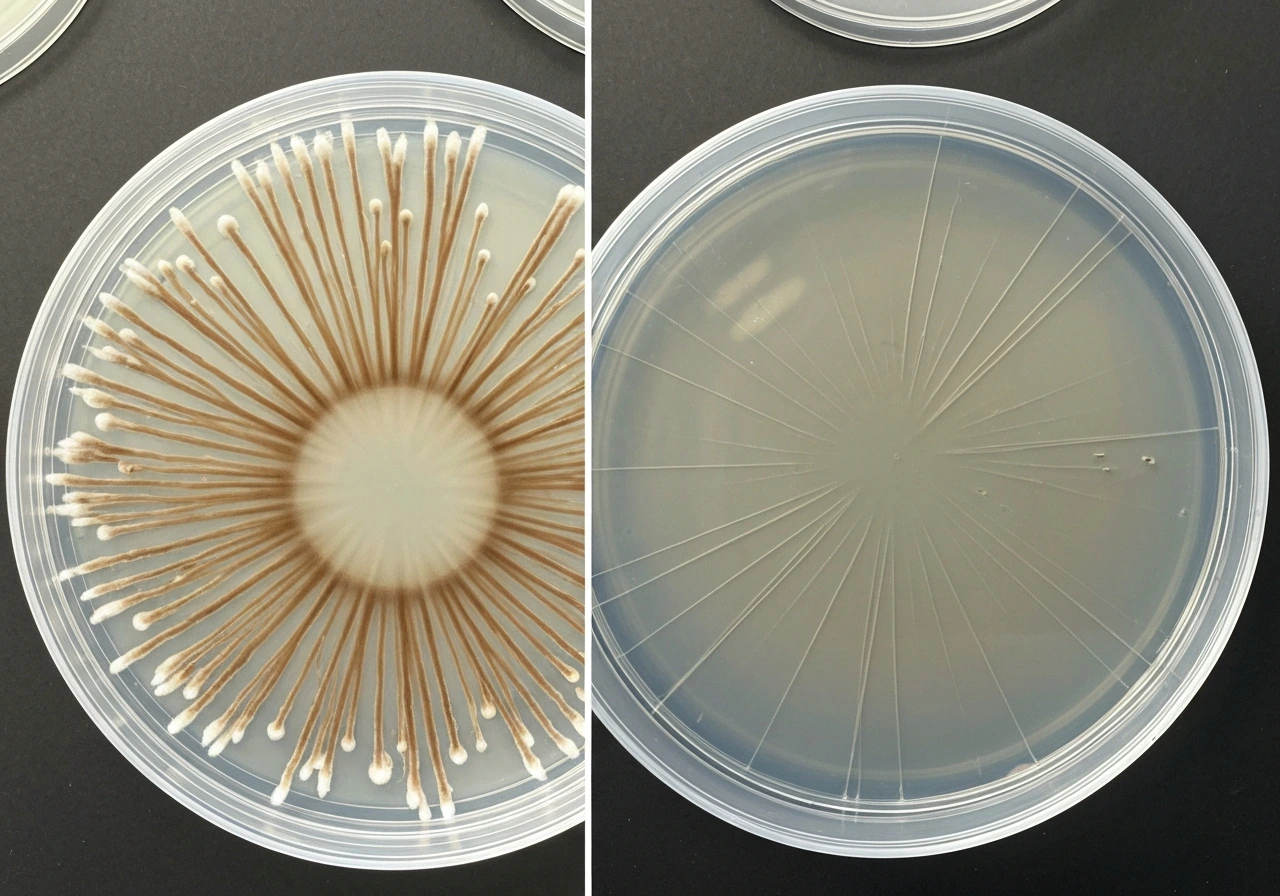
Growth rate varies enormously by species, substrate, and conditions. On laboratory agar, many saprotrophic fungi extend radially at somewhere between 0.2 and 10.8 millimeters per day. Grey oyster mushroom (Pleurotus ostreatus) can reach 8 mm per day on ideal culture medium in the early growth phase, and some Pleurotus strains on optimized media have shown radial extension rates approaching 17 mm per day. On real-world substrates those numbers drop. A general benchmark: for most commonly cultivated species, full colonization of an inoculated substrate takes two to five weeks under optimal conditions, and first visible mycelial strands often appear within three to seven days of inoculation.
What limits those rates? Several things act simultaneously. Nutrient diffusion is one: the mycelium has to enzymatically break down substrate ahead of its tips, and that process has a speed limit set by enzyme kinetics and diffusion physics. Substrate porosity and stiffness matter too: research published in 2024 specifically found that substrate stiffness can be used to tune mycelium growth rates, and denser or less porous substrates slow penetration. Water availability sets a hard floor. Oxygen diffusion limits how deep into a substrate the network can remain metabolically active. And space itself eventually becomes a constraint: in a closed jar or block, the mycelium consumes available nutrients and runs out of room to expand.
This is actually a theme worth pausing on. How growth works across living systems consistently comes back to the same idea: growth is not unlimited, it is bounded by resource supply, physical structure, and the organism's own metabolic constraints. Mycelium is a perfect illustration of that principle operating in real time.
Why your mycelium might have stalled (and how to fix it)
If growth has slowed to almost nothing or stopped entirely, run through this checklist systematically before assuming the culture is dead.
| Problem | What you will observe | Fix |
|---|---|---|
| Too dry | Thin, wispy growth that stops advancing; substrate pulling away from container walls | Increase relative humidity to 80–85%; mist lightly but avoid pooling water |
| Too wet / waterlogged | Yellow or brown patches; sour smell; slow growth even with signs of life | Improve drainage; reduce misting; check substrate field capacity before inoculation |
| Poor aeration / CO2 buildup | Dense colonization near air exchange points but stalling in the center or base | Add or enlarge filter patches; fan briefly 1–2x daily; avoid fully sealed containers |
| Temperature too low or too high | Dramatically slowed extension; in heat cases, yellow or dying sectors | Bring temperature into the 21–27°C range for most species; check with a probe thermometer placed at substrate level |
| Wrong pH | Patchy or no colonization on otherwise suitable substrate | Test substrate pH; aim for 5.5–7.0; acidify with gypsum or coffee grounds, or alkalify cautiously with lime |
| Nutrient-poor substrate | Very slow colonization; pale, thin mycelium | Switch to or supplement with a richer substrate (add wheat bran, soy hulls, or another nitrogen supplement at 10–20% by weight) |
| Contamination | Green, black, pink, or orange patches; sour or sweet off-odors | Remove contaminated substrate immediately; do not attempt to save it; sterilize containers; review inoculation hygiene |
Contamination deserves a special note. Competing molds and bacteria almost always outcompete mycelium when conditions are already suboptimal. If you are seeing contamination repeatedly, the root cause is usually inadequate sterilization or pasteurization of the substrate, poor inoculation hygiene, or environmental conditions that favor competitors (typically too warm, too wet, or too much nitrogen relative to carbon). Fix the conditions first, and contamination rates drop significantly.
Practical steps to encourage healthy mycelium growth starting today
Whether you are cultivating edible mushrooms, studying fungal biology, or trying to understand what is growing in your compost or walls, the core actions are the same: get the five environmental factors right and remove physical barriers to growth.
- Choose the right substrate for your target species. Hardwood sawdust supplemented with wheat bran works well for most gourmet species. Straw is simpler and more forgiving for oyster mushrooms. Match the substrate to the species' natural ecological role.
- Hydrate the substrate to field capacity: it should hold its shape when squeezed but release only a few drops of water. Over-wet substrate is one of the most common growth killers.
- Inoculate with a healthy, active spawn and minimize contamination risk by working in a still-air or clean environment. Speed matters at this stage.
- Maintain temperature between 21 and 27°C during colonization. Use a thermometer at substrate level, not just room air temperature.
- Ensure CO2 can escape. A simple polyfill filter patch on a jar lid, or small holes covered with micropore tape, is enough for most small-scale setups.
- Keep relative humidity around 80 to 85% during colonization by lightly misting the environment (not the substrate directly) and covering loosely.
- Once colonization is complete, switch to fruiting conditions: drop temperature, increase fresh air exchange, raise humidity to 90–95%, and introduce indirect light.
If you are approaching this from a nature observation angle rather than cultivation, compost is one of the best places to watch mycelium at work. Turning a backyard compost pile regularly exposes the white thread networks running through partially decomposed organic matter. You are watching the fungus eat in real time. Those threads are breaking down cellulose and lignin, releasing nutrients back into the soil, and doing the same biochemical work that wood-decay fungi do in forests.
A quick comparison of common growth substrates
If you are deciding where to start, this comparison covers the most accessible options and what to realistically expect from each one.
| Substrate | Best for | Colonization speed | Contamination risk | Notes |
|---|---|---|---|---|
| Hardwood sawdust + wheat bran | Oyster, shiitake, lion's mane | Fast (2–3 weeks) | Medium (requires sterilization) | High yield potential; requires pressure cooking |
| Straw (pasteurized) | Oyster mushrooms | Fast (1–2 weeks) | Low-medium | Cheap and widely available; pasteurize at 70–80°C, not full sterilization |
| Brown rice flour + vermiculite (BRF tek) | Cubensis and other gourmet species | Moderate (2–4 weeks) | Medium | Beginner-friendly small-scale method |
| Agar (laboratory) | Culture work, cloning, testing | Very fast (2–5 days visible) | Low with proper technique | Best for observation and stock culture maintenance |
| Compost / decaying wood (outdoor) | Wild species, mycorrhizal experiments | Variable | Very low (open system) | Not controllable but great for observation |
| Cardboard (used coffee grounds on top) | Oyster mushrooms, informal trials | Slow | Medium-high | Low-cost experiment; expect variable results |
For beginners, pasteurized straw inoculated with oyster mushroom spawn is the most forgiving starting point. For more controlled observation of hyphal growth patterns, agar plates give you something close to what a lab sees. The choice of substrate is not just logistical: it is one of the main dials controlling how fast your mycelium grows and how healthy it stays.
The bigger picture: mycelium as a model for bounded growth
Mycelium growth is a striking example of how living systems expand efficiently within real constraints. The tip-growth-and-branching strategy lets fungi cover large areas and penetrate complex substrates without requiring a centralized control system. Every branch tip is semi-autonomous, extending based on local chemical gradients and physical resistance. Yet the whole network integrates and transports resources across the entire structure.
That same pattern, growth that is directional, resource-dependent, and physically constrained, shows up across biology. Consider how an apple develops on its tree, relying on vascular transport networks and resource allocation from the parent plant, or how a fig grows through an intimate relationship with external organisms that mirrors the mycorrhizal exchange. Even date palms grow by concentrating resources in ways governed by physical transport limits. Growth, wherever it occurs, is always a negotiation between what the organism is trying to do and what the environment and physics allow.
Mycelium just makes that negotiation unusually visible. You can watch it happen in a jar on your kitchen counter, spreading a few millimeters per day, problem-solving its way through a block of sawdust, and pausing every time you get one of those five environmental factors wrong. That makes it one of the best hands-on models for understanding how growth actually works.
FAQ
How does mycelium grow faster, and what should I change first?
Start with the one limiting factor, usually water activity and fresh air exchange. If it is too dry, hyphal tips lose turgor and growth stalls, but if it is too wet you may block oxygen diffusion. In practice, adjust moisture and then verify you are not sealing the block too tightly (CO2 buildup slows extension).
Is it normal for mycelium to look patchy or uneven during colonization?
Yes, early colonization often spreads in uneven fronts because the network follows local nutrient and moisture gradients. However, if patchiness persists after the substrate should be mostly colonized, it can indicate poor sterilization, excessive nitrogen, or airflow that is too low or too high (causing drying).
Can mycelium grow without light?
Most species colonize normally in the dark because tip growth is mainly driven by water, oxygen, temperature, and substrate chemistry. Light is more likely to affect the transition to primordia in some species, so lack of light usually impacts fruiting, not initial spread.
What is the difference between water content and water activity (aw), and why does it matter?
Water content is how much moisture is present, water activity (aw) reflects how available that water is to microbes. Two substrates can have the same moisture level but different aw values, leading to very different colonization performance, especially near the lower growth threshold.
Why does mycelium stop growing even though the substrate still looks full of nutrients?
Common causes are oxygen limitation (CO2 and low O2 in dense, wet, or sealed setups), temperature outside the species window, or pH that shuts down enzyme function at the hyphal tip. Nutrient diffusion limits can also slow progress if the substrate is very dense or poorly hydrated.
Does mycelium always grow on the surface first, or does it penetrate right away?
Many substrates allow rapid penetration once hyphae locate accessible pits or microchannels. On cellulose-rich or wood-like materials, outward spread can be visible before full internal colonization, but penetration depends heavily on substrate structure and porosity.
How can I tell contamination versus slow colonization from the appearance alone?
Mycelium typically forms consistent, fuzzy growth with a relatively uniform texture, while many contaminants produce distinct colors, sudden spots, slimy regions, or strong odor. If growth ceases and you see rapid color changes or wet surfaces, treat it as contamination rather than “just a slow strain.”
If contamination keeps coming back, what is the most likely mistake?
Often it is hygiene and process control: inadequate sterilization or pasteurization, contaminated inoculum, or handling during inoculation. Environmental favoritism matters too, but repeated failures usually trace to substrate prep and inoculation technique more than to temperature alone.
How long should it take for mycelium to fully colonize a substrate?
A practical benchmark for many cultivated species is about two to five weeks under good conditions, with first visible growth often appearing within a few days. If you are dramatically outside that window, re-check moisture, temperature, and fresh air first before assuming the spawn is dead.
Why does mycelium colonize, but mushrooms never fruit?
Colonization and fruiting require different triggers. Even with a fully colonized block, fruiting can fail if you have not changed conditions (often temperature shift, increased fresh air, higher humidity, and sometimes light depending on species). Also note that conditions that support colonization can reduce porosity or oxygen transfer needed for developing fruit bodies.
Does substrate choice affect not just how fast mycelium grows, but how much it fruits?
Yes. Substrate composition changes enzyme matching and nutrient availability, which affects both colonization speed and later resource allocation for fruiting. If nutrition is skewed (for example, too much nitrogen relative to carbon), colonization may look fine but contamination risk and fruiting performance can suffer.
Can I speed up growth by increasing temperature or humidity beyond the recommended range?
Usually not. Higher temperature can denature proteins and promote competitors, and excess humidity can waterlog the substrate, reducing porosity and oxygen diffusion. The better approach is to stay within the species-specific window and make targeted adjustments.
What role does branching play in how quickly mycelium covers an area?
Branching creates additional tips, and each tip becomes a new growth point. Faster area coverage often correlates with healthy tip growth plus regular initiation of new branches, which depends on adequate water, oxygen, and correct pH and substrate chemistry.
How Does It Grow: Biological and Crystal Growth Explained
Step-by-step guide to how living cells and crystals grow, including conditions, limits, slowdown causes, and troubleshoo


