Cells increase in number by dividing. One cell splits into two through a tightly choreographed process called the cell cycle, which ends with mitosis (the nucleus divides) and cytokinesis (the cytoplasm splits). That's the core answer. Everything else on this page explains exactly how that works, what conditions have to be in place for it to happen, and what stops cells from dividing forever.
Cells Grow in Number by Doing What? Cell Division Steps

Why cells need to divide to grow in number
Here's the fundamental idea: cells can't just appear from nothing, and they can't bud off pieces of themselves randomly. The only way to make more cells is to take one existing cell and replicate everything inside it, then physically split it into two complete, functional daughter cells. This is called cell division, and it's how every tissue in your body expands, how a wound heals, and how a single fertilized egg eventually becomes trillions of cells. The process is the same whether we're talking about skin cells, liver cells, or the cells lining your gut.
The formal term for an increase in cell number is hyperplasia. That's worth knowing because it sits right next to a term that gets confused with it constantly: hypertrophy, which means an increase in cell size. More on that distinction shortly. For now, the key point is that hyperplasia (more cells) only happens through division.
Cell division is the direct mechanism: mitosis and cytokinesis
In eukaryotic cells (which includes everything from yeast to human cells), division has two main events that happen in sequence. First, the nucleus divides in a process called mitosis, distributing one complete copy of the DNA to each side of the cell. Then cytokinesis physically cleaves the cytoplasm in two, sealing the deal with two separate daughter cells. Neither step alone is enough. You need both.
The four phases of mitosis
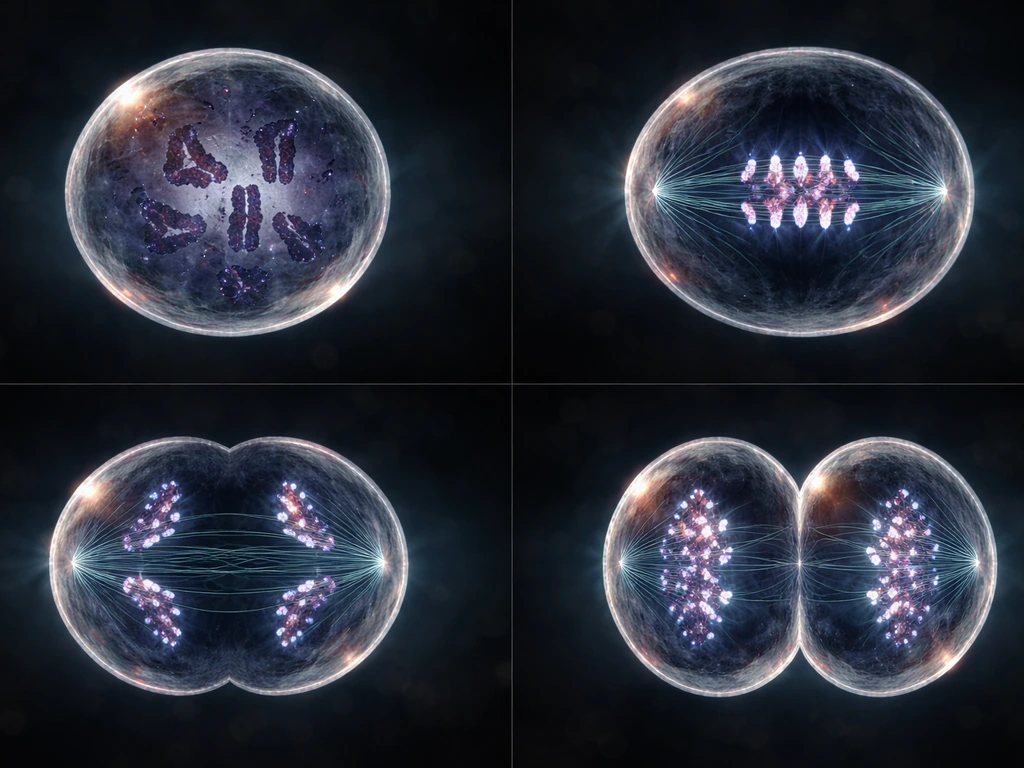
Mitosis itself moves through four phases, each doing a specific job to make sure the chromosomes end up separated correctly.
- Prophase: Chromosomes condense and become visible. The mitotic spindle starts forming, and the nuclear envelope begins to break down.
- Metaphase: Chromosomes line up along the middle of the cell (the metaphase plate). Spindle fibers from opposite poles attach to each chromosome. This is the cell doing a quality check before committing.
- Anaphase: Sister chromatids are pulled apart toward opposite poles of the cell. This is the moment of actual separation.
- Telophase: The chromosomes arrive at opposite poles, and a new nuclear envelope forms around each set. The cell now has two nuclei.
Cytokinesis typically begins before telophase is fully complete. In animal cells, a contractile ring of proteins pinches the cell membrane inward until the two daughter cells are fully pinched off. In plant cells, a cell plate forms between the two nuclei and builds outward into a new cell wall. Either way, the result is two genetically identical daughter cells, each with its own nucleus and a full complement of DNA.
The full picture: the cell cycle leading up to division
Mitosis and cytokinesis are the finale, but they only work because of everything that happens beforehand. The complete sequence is called the cell cycle, and it has three main stages before division even begins: G1, S phase, and G2 (collectively called interphase), followed by the M phase (mitosis plus cytokinesis).
- G1 (first gap): The cell grows in size, synthesizes proteins, and checks whether conditions are favorable for division. Think of it as the preparation phase.
- S phase (synthesis): DNA replication happens here. Every chromosome is copied so that each daughter cell will get a complete genome. The centrosome also duplicates during this phase.
- G2 (second gap): The cell continues to grow and produce proteins needed for mitosis. Another round of checks happens to confirm DNA replication completed without errors.
- M phase: Mitosis followed by cytokinesis. This is where the actual increase in cell number happens.
The G1 and G2 gap phases exist specifically to give cells time to grow and double-check their work. They're not dead time. A cell that skips those checks risks dividing with incomplete or damaged DNA, which is exactly the kind of mistake that causes problems down the line.
Growing in number vs. growing in size: not the same thing
This is a distinction that trips up a lot of students, so it's worth being direct about it. Cells can get bigger without dividing at all. Hypertrophy (larger cell size) and hyperplasia (more cells) are two completely different things, even though both result in tissue that looks or functions differently than it did before.
| Term | What happens | Cell number | Cell size | Example |
|---|---|---|---|---|
| Hypertrophy | Individual cells enlarge | Stays the same | Increases | Muscle cells growing after weight training |
| Hyperplasia | Cells divide to produce more cells | Increases | Often similar to parent cell | Skin cells replacing a scrape |
| Both together | Cells divide AND enlarge | Increases | Increases | Normal organ development in embryos |
When someone asks how cells grow in number, the answer is always hyperplasia through division, not hypertrophy. A muscle that gets stronger through training is largely doing hypertrophy, not making dramatically more muscle cells. A cut on your skin heals through hyperplasia, with skin cells actively dividing to fill the gap. The distinction matters practically because the two processes are controlled by different signals and have different implications for health.
It's also worth noting that cell size and cell number can change independently depending on the tissue type. Some cells, like neurons, rarely divide at all after development and instead grow very long projections to extend their reach. If you're curious how this plays out in specific cell types, the biology of animal cells and skin cells offer interesting examples of how growth strategies vary.
What a cell actually needs before it can divide
Division isn't automatic. A cell requires a specific set of conditions before it will commit to the cell cycle. Think of it like baking bread: you can have the recipe, but if you're missing yeast, the right temperature, or enough flour, nothing rises. Cells are similar.
Nutrients and energy
Dividing takes a serious investment of resources. The cell needs to replicate its entire DNA, manufacture a full set of organelles, build proteins for the spindle, and physically split itself in two. All of that requires ATP (the cell's energy currency), raw building blocks like amino acids and nucleotides, and a steady supply of nutrients. Starve a cell of glucose or essential amino acids and it will typically pause in G1 rather than proceed into S phase.
Growth signals and hormones
Cells don't just divide whenever they feel like it. In a multicellular organism, division is regulated by extracellular signals. Mitogens are signaling molecules that push cells to enter or progress through the cell cycle by relieving internal inhibitory controls. Growth factors, which are a related but distinct category, specifically stimulate the cell to grow larger and increase its protein production. Without the right signals arriving at the right receptors, most cells in your body stay put in a resting state called G0. Hormones, cytokines, and locally produced proteins all contribute to this signaling environment.
Space and physical context
Cells also need physical room and the right attachment context. Many cell types require attachment to the extracellular matrix (ECM) to survive and divide. When cells lose that contact, they trigger a form of programmed cell death called anoikis. This is one of the body's ways of preventing cells from proliferating in places they don't belong. Adequate space in the surrounding tissue also matters: cells that are crowded by neighbors receive inhibitory signals that slow or stop division, a phenomenon covered in the next section.
Waste removal
Cells actively produce metabolic waste during all the energy-intensive work of growing and dividing. If waste products build up and the local environment becomes toxic or imbalanced, division stalls. This is part of why blood flow and lymphatic drainage matter so much for growing tissues: they supply nutrients and carry waste away, keeping conditions favorable.
The limits: checkpoints, contact inhibition, and what happens when they fail
One of the most important things to understand about cell division is that the cell cycle has built-in brakes. These aren't optional extras; they're fundamental to why your body doesn't grow uncontrollably. There are three main layers of control worth knowing.
Cell-cycle checkpoints
The cell cycle has specific decision points called checkpoints where the cell pauses and evaluates its situation before moving forward. The G1/S checkpoint is the most critical: if conditions aren't right (nutrients low, DNA damaged, signals absent), the cell is blocked from entering S phase. Proteins called cyclin-dependent kinases (Cdks) drive the cell through each transition, but they only work when paired with their matching cyclin proteins and when inhibitory proteins like p21 are not active. DNA damage triggers the tumor suppressor p53 to activate p21, which blocks Cdk activity and arrests the cell cycle. The G2/M checkpoint does a similar job before mitosis begins, and the spindle assembly checkpoint inside mitosis itself delays chromosome separation until every chromosome is properly attached to the spindle. These are not redundant systems; they're sequential filters.
Contact inhibition
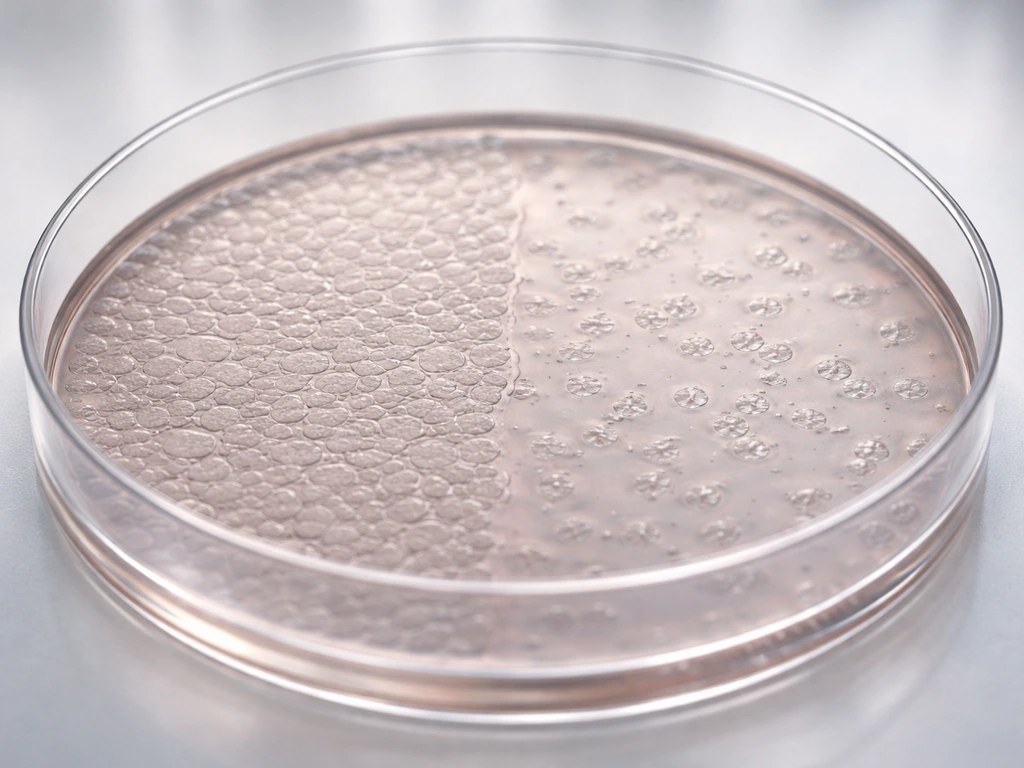
Normal cells stop dividing when they run out of space and make contact with surrounding cells. This is called contact inhibition. It's easy to observe in a lab: grow normal cells in a dish and they form a neat single layer, then stop. Remove some cells, and the neighbors divide to fill the gap, then stop again. The mechanism involves inhibitory signals exchanged between touching cells that suppress cell-cycle progression. It's one of the body's elegant ways of matching cell number to available space.
Apoptosis: the built-in off switch
When a cell is severely damaged, in the wrong location, or receiving signals that it's no longer needed, it can activate a programmed self-destruction sequence called apoptosis. This isn't cell death from injury; it's an organized, controlled process that the cell runs on itself. Apoptosis removes dangerous or unnecessary cells without triggering inflammation, and it plays a huge role in normal development (it's how fingers separate in the embryo, for instance).
When the limits fail: cancer
Cancer is essentially what happens when these control systems break down. Tumor cells often ignore mitogen signals (or produce their own), lose sensitivity to contact inhibition, bypass checkpoint controls, and fail to undergo apoptosis even when conditions should trigger it. The result is uncontrolled hyperplasia: cells dividing when and where they shouldn't, accumulating mutations along the way, and eventually invading tissue they don't belong in. Key proteins involved in this breakdown include mutated forms of p53 (present in a huge fraction of human cancers), overactive CDK4/6-cyclin D complexes that push cells through G1 regardless of conditions, and defects in the retinoblastoma (RB) protein that normally acts as a brake on G1 progression. Understanding normal division regulation makes it clear why these specific mutations are so consistently dangerous: each one removes a layer of the control system that evolution spent a very long time building.
The biology of cell growth and division also raises interesting questions about how different cell types manage this process. Red blood cells, for example, handle division and longevity in a genuinely unusual way. And the rate at which cells complete the full cycle varies dramatically depending on cell type and organism, which connects directly to how fast tissues can grow or repair themselves. The answer to how fast do cells grow depends largely on how quickly cells complete their cell cycle and proceed through checkpoints rate at which cells complete the full cycle.
Putting it all together
Cells grow in number by dividing, and they divide through a coordinated sequence: interphase (G1 growth, S-phase DNA replication, G2 preparation) followed by mitosis (prophase, metaphase, anaphase, telophase) and then cytokinesis. Every step requires the right inputs (nutrients, energy, signals, space) and passes through quality-control checkpoints. When the system runs correctly, you get precise, regulated production of new cells exactly where and when they're needed. When it doesn't, you get disease. That's the whole story of how cell number increases, from the molecular machinery up to the organism level. In animal cells specifically, this growth in number depends on the cell cycle and on signals that determine when division is allowed.
FAQ
So if cells get bigger, does that mean they are growing in number?
They can increase in size without increasing in number, which is hypertrophy. In practice, if a tissue looks larger because individual cells are bigger (for example, muscle after training), that is not the same mechanism as hyperplasia, which requires complete cell division into two daughter cells.
Does cell number increase with mitosis alone?
In most textbooks, the phrase “cell division” refers to the full outcome of making two daughter cells, which requires both mitosis and cytokinesis. If you only have nuclear division (mitosis) without cytoplasm splitting, you do not end with two separate daughter cells and the cell number does not increase.
If a cell starts the cell cycle, does it always end up producing two new cells?
Cells usually only commit to division when external and internal conditions pass a checkpoint, especially the G1/S checkpoint. If a cell enters S phase with damaged DNA, checkpoints can trigger arrest or programmed removal, so “beginning to divide” is not the same as “successfully increasing cell number.”
How can the speed of tissue growth change if the steps of the cell cycle are the same?
Yes, by controlling how many cells enter the cycle, how fast they progress through checkpoints, and whether division is completed. A change in tissue growth rate can happen even if the same cell type divides, by shifting nutrient availability, mitogen levels, or checkpoint timing.
What happens to cell division when cells are crowded, and does it always work the same way in every tissue?
Contact inhibition is mainly about whether cells receive inhibitory signals when they are too crowded or when they have extensive cell-to-cell contact. But some cell types can be unusual, and in cancer this control often fails because tumor cells ignore those inhibitory cues.
Can a detached cell still divide if nutrients and signals are present?
No, if key attachments are missing, many cells activate anoikis instead of dividing. This is especially relevant for epithelial and other cells that depend on extracellular matrix attachment to survive and proceed through the cycle.
Are there cell types that don’t increase in number by dividing?
Yes. Red blood cells in mammals do not divide after maturation, yet they survive for days to weeks depending on conditions. This is a useful exception because it shows that cell population size in tissues is not always driven by ongoing division.
When checkpoints stop a cell, does it die or just pause?
If a cell cycle checkpoint arrests the cell, it may repair damage and later re-enter, or it may remain in a resting state like G0. In other cases, severe damage can lead to apoptosis, which removes the cell instead of increasing cell number.
How do checkpoints actually prevent uncontrolled division at the molecular level?
Cyclins and Cdks are regulated so a cell cannot progress just because it has enough energy. For example, DNA damage can activate p53, which increases p21 and inhibits Cdk activity, preventing entry into or progression through later phases until the problem is resolved.
What specifically goes wrong in cancer to cause increased cell number where it should not occur?
In cancer, cells often bypass one or more layers of control, such as checkpoint arrest, sensitivity to contact inhibition, or apoptosis triggers. The key point is that uncontrolled hyperplasia results from losing these constraints, not from dividing faster under normal regulation.
How Do Animal Cells Grow: Cell Cycle, Signals, Limits
How animal cells grow: cell cycle checkpoints, growth signals, nutrients, internal conditions, and limits like contact i