A neuron grows by doing something most cells never attempt: it shoots out a long, thin cable called an axon, sometimes stretching more than a meter in the human body, while simultaneously sprouting shorter branching arms called dendrites. That growth isn't random sprawl. It follows a tight biological script, with each stage driven by specific molecular machinery and reined in by equally specific brakes. Here's how the whole process works, from the moment a neuron is born to the moment it locks into a working circuit.
How Do Neurons Grow Step by Step From Outgrowth to Synapses
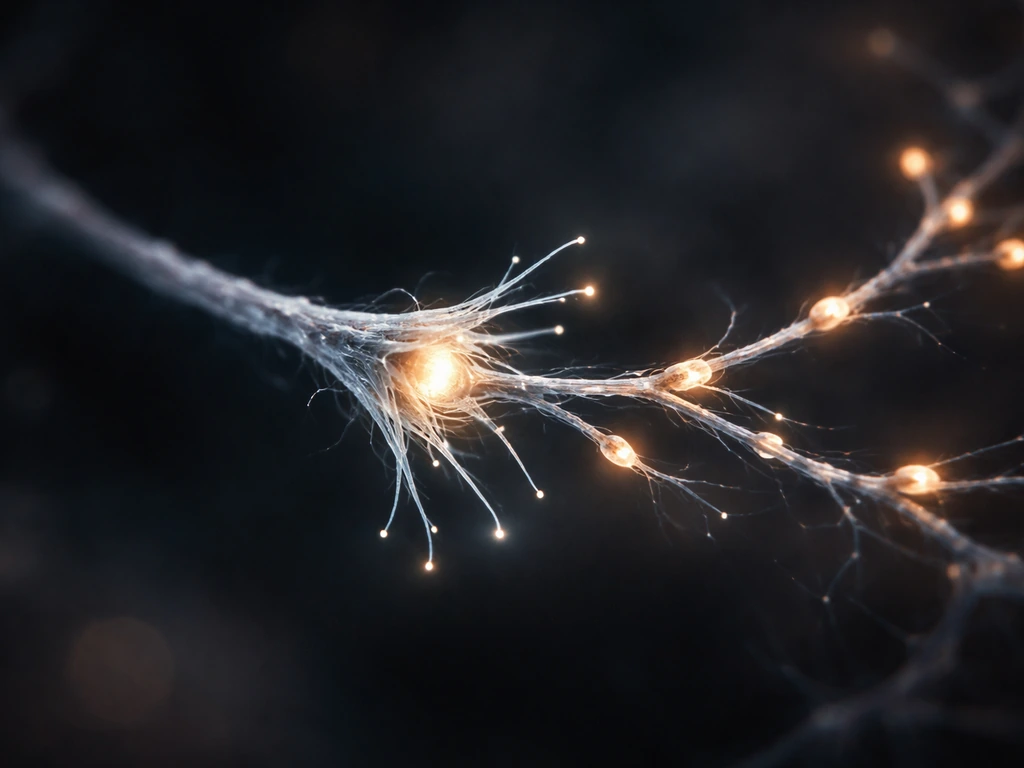
Neuronal growth basics: structure, stages, and scales
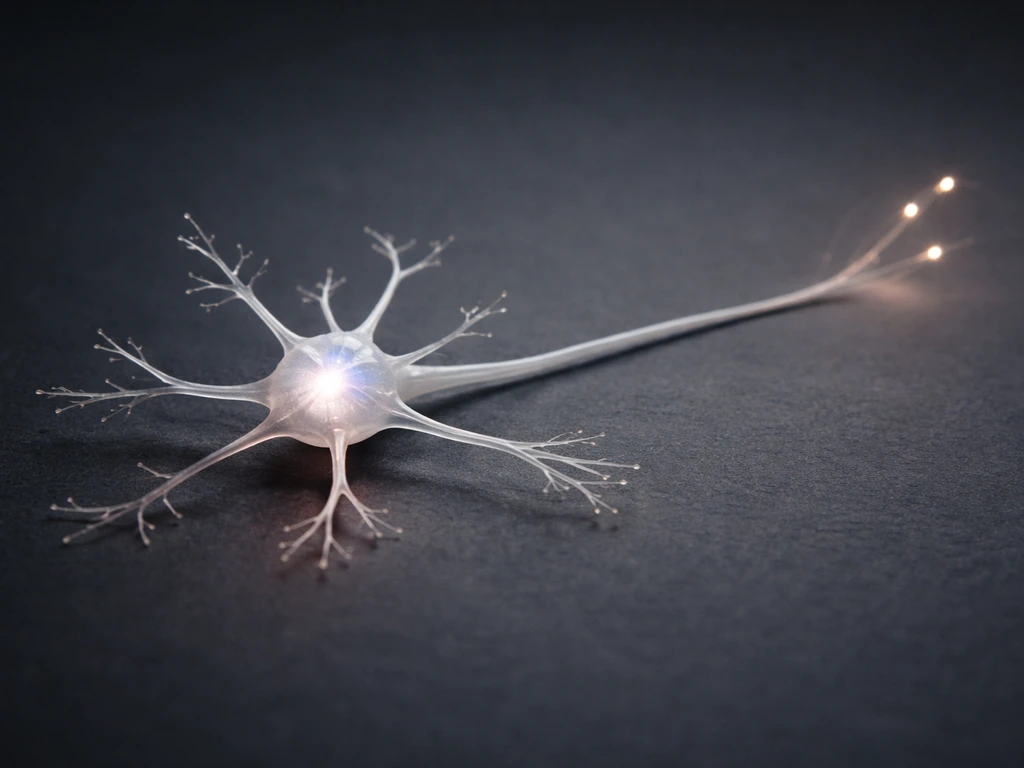
To understand how neurons grow, you first need a clear picture of what they're actually building. A typical neuron has three main parts: the cell body (soma), which houses the nucleus and most of the metabolic machinery; dendrites, which are relatively short, branching processes that receive incoming signals; and one axon, a single long projection that transmits action potentials away from the soma toward other neurons or target tissues. The axon initial segment, the stretch between the axon hillock and the first unmyelinated region, is where action potentials actually fire, thanks to a dense cluster of voltage-gated channels anchored by scaffold proteins.
Growth happens in overlapping stages. First, the newborn neuron migrates to its final position in the nervous system. Then it extends a single axon and several dendrites (process outgrowth). The axon pathfinds to its target, synapses form where contacts are made, and finally the circuit is refined by activity-dependent pruning. As these mechanisms unfold, the nervous system changes and grows by reshaping connections through activity, guidance, and pruning how does the nervous system change and grow. Each stage has its own molecular drivers and, just as importantly, its own constraints. The scales involved are striking: human cortical axons can be 1 meter long, while a single dendritic arbor may carry thousands of individual synaptic spines.
Guiding principles: polarity, migration, and targeting
Before a neuron can grow, it has to know which way is which. Establishing polarity, deciding which process becomes the axon and which become dendrites, is one of the first critical decisions. This is driven by an asymmetric distribution of signaling proteins inside the young cell. PI3-kinase activity, for instance, concentrates at the tip of the future axon, while PTEN (its antagonist) stays distributed elsewhere. Once one neurite wins this molecular competition and commits to becoming the axon, the rest are suppressed from doing the same. It's a winner-take-all arrangement.
Migration comes before or alongside polarity in many brain regions. Neurons born in proliferative zones like the ventricular zone in the cortex must travel, sometimes long distances, to reach their final layer. They do this by crawling along radial glia fibers, guided by chemical gradients including Reelin, a secreted glycoprotein that tells neurons when to stop and detach. Get the migration signal wrong and neurons end up in the wrong layer, which is exactly what happens in certain cortical malformations. Think of it like a GPS route: the destination is set before the car leaves the driveway.
Targeting, getting the axon to the right neighborhood and then the right cell, involves a separate but overlapping set of molecular instructions. Axons are sorted into broad regions by graded signals (like ephrin gradients in the visual system), then refined by local cues and activity. The nervous system essentially uses a two-step zip code: a rough area code from gradients, and a precise address from activity and local signaling.
How axons extend: growth cones, guidance cues, and pathfinding

The growth cone is one of biology's most fascinating structures. It sits at the tip of every growing axon (and growing dendrite) like a tiny hand with constantly probing fingers, called filopodia, and flat sheet-like extensions called lamellipodia. The growth cone crawls through tissue, sampling the environment with surface receptors and translating what it finds into directional movement. Speed is usually in the range of 1 to 10 micrometers per hour for axons navigating tissue in vivo, though it can be faster in culture on permissive substrates.
Guidance cues come in four flavors: attractive short-range, repulsive short-range, attractive long-range (diffusible), and repulsive long-range. Key molecular families include netrins (which can attract or repel depending on the receptor), semaphorins (mostly repulsive), ephrins (repulsive, important for topographic mapping), and slits (repulsive, keeping axons out of the midline once they've crossed). The growth cone integrates these signals simultaneously, like a navigator reading multiple maps at once. When an axon reaches a choice point, like the midline of the spinal cord, it switches its receptor expression to become repelled by signals it was previously attracted to, ensuring it doesn't cross back.
Inside the growth cone, the cytoskeleton is doing the actual mechanical work. Actin polymerization at the leading edge pushes filopodia forward, while myosin-driven retrograde flow pulls actin rearward. When a guidance receptor engages an attractive cue, it locally stabilizes actin and reduces retrograde flow, allowing the leading edge to advance in that direction. Repulsive cues do the opposite, causing local actin collapse. Microtubules then consolidate advances made by actin, extending into the growth cone and eventually becoming the core of the new axon shaft. It's a beautifully coordinated ratchet system.
How dendrites and synapses form: activity, plasticity, and pruning
While the axon is pathfinding, dendrites are doing their own kind of growing. Dendritic growth is less about long-range navigation and more about local branching and elaboration. Young dendrites sprout from the soma, branch repeatedly, and eventually develop spines, tiny protrusions that are the physical sites of most excitatory synapses. The shape and density of dendritic spines matter enormously: thin, motile spines are immature and plastic, while mushroom-shaped spines with large heads are mature and stable.
Synapses don't form randomly. When an axon terminal contacts a dendrite and the contact is reinforced by correlated activity (both cells firing together), molecular signals stabilize the synapse. The presynaptic terminal recruits synaptic vesicle machinery; the postsynaptic dendrite clusters glutamate receptors and scaffold proteins like PSD-95. This is Hebbian plasticity in its most literal, structural form: synapses that are active together wire together. Synapse formation also involves trans-synaptic organizing proteins like neurexins and neuroligins, which physically bridge the two sides of the synapse and initiate the recruitment of the synaptic machinery on both ends.
Growth doesn't just mean more. A critical and often underappreciated part of neural development is pruning. The brain initially overproduces synapses and dendritic branches, then eliminates the ones that aren't used or reinforced. In humans, synaptic pruning is especially active during childhood and adolescence. Microglia, the brain's resident immune cells, physically engulf and remove weak or inactive synapses, guided by complement proteins that tag synapses for elimination. So the final wiring of a circuit reflects not just what was added, but what survived competition.
The cellular mechanics: cytoskeleton, transport, and membrane expansion
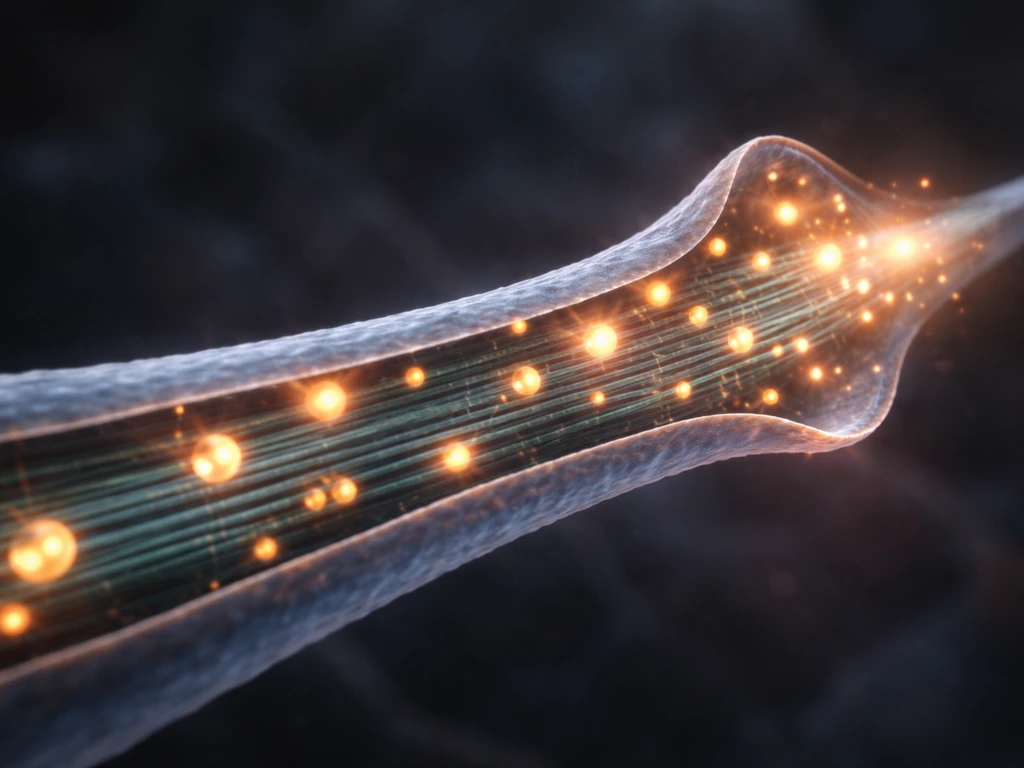
Growing an axon a meter long is a logistical problem at least as much as a molecular one. The cell body has to continuously supply the growing tip with membrane, cytoskeletal components, organelles, and signaling molecules. This happens via axonal transport, a system of molecular motor proteins (kinesins moving cargo toward the axon tip, dyneins moving cargo back toward the soma) walking along microtubule tracks. Mitochondria have to be positioned at high-energy-demand sites, especially at growth cones and active synapses. Failure in axonal transport is a central feature of many neurodegenerative diseases.
Microtubule polarity is a key organizing principle here. In axons, microtubules are oriented with their plus ends pointing toward the tip, which allows kinesin motors to efficiently deliver cargo outward. In dendrites, microtubule polarity is mixed, with both plus-end-out and minus-end-out orientations, and this difference helps sort which cargoes go into axons versus dendrites. Disrupting microtubule polarity scrambles cargo delivery and disrupts neuronal identity.
Membrane expansion at the growth cone is not just passive stretching. The cell actively delivers membrane vesicles by exocytosis at the leading edge of the growth cone and recycles membrane from the trailing edge by endocytosis. This membrane flow is coupled to cytoskeletal dynamics and is regulated by small GTPases like Rac1, Cdc42, and RhoA. RhoA activation causes growth cone collapse (retraction), while Rac1 and Cdc42 promote outgrowth. Many guidance cues ultimately work by activating or inhibiting these GTPases.
Growth constraints and limits: energy, space, signaling balance, and competition
Neurons don't keep growing forever, and the reasons are both physical and molecular. The brain is enclosed in a rigid skull, so total volume is a hard constraint. But the limits on individual neuron growth are more nuanced and, honestly, more interesting than just running out of room.
Energy is a major bottleneck. Neurons are among the most metabolically expensive cells in the body. Growing an axon requires ATP for polymerizing actin and tubulin, running motor proteins, maintaining ion gradients, and driving membrane trafficking. The metabolic cost of maintaining a long axon is substantial, which is part of why axon diameter and myelination are so tightly regulated. Axons that are too thin for their length would simply fail to maintain themselves energetically.
Signaling balance is another major brake. Growth-promoting signals like neurotrophins (BDNF, NGF) are produced in limited quantities by target tissues. Axons compete for these trophic signals, and neurons that fail to capture enough undergo apoptosis. This is not a design flaw but a feature: neurotrophic competition ensures that the right number of neurons wire to each target and that excess neurons are eliminated. It's a market economy for survival signals.
The extracellular matrix also plays a gating role. Chondroitin sulfate proteoglycans (CSPGs) accumulate around mature synapses as perineuronal nets, physically restricting structural plasticity. Studies have shown that digesting CSPGs with the enzyme chondroitinase ABC restores spine motility and synaptic remodeling in adult tissue, revealing that the ECM is actively holding growth in check in the mature brain. Growth doesn't just stop because neurons forget how to grow; the environment actively suppresses it.
Finally, there's competitive pruning at the level of axon terminals. Where multiple axons initially innervate a single target cell, activity-dependent competition eventually leads to one axon dominating and the others withdrawing. The neuromuscular junction is the classic example: each muscle fiber is initially innervated by multiple motor axons, and through competitive activity-dependent signaling, it ends up with just one. Similar processes refine connections throughout the nervous system.
What supports healthy neuron growth: practical factors in culture and in the brain
If you're working with neurons in culture or studying neuronal growth in a developmental or regenerative context, the factors that matter most are fairly well-established. Here's what the evidence points to:
- Substrate adhesion molecules: laminin, fibronectin, and poly-D-lysine coatings dramatically improve axon outgrowth in culture by providing integrin-binding sites that anchor the growth cone and promote forward movement.
- Neurotrophic factors: BDNF, NGF, NT-3, and GDNF promote survival and process outgrowth in a cell-type-specific way. BDNF is especially potent for cortical and hippocampal neurons; NGF is critical for peripheral sensory and sympathetic neurons.
- Removing inhibitory signals: CSPGs, myelin-associated glycoprotein (MAG), and Nogo are major inhibitors of axon growth in the adult CNS. Blocking or digesting them (e.g., with chondroitinase) substantially increases regrowth after injury.
- Activity and depolarization: moderate neuronal activity promotes dendrite elaboration and synapse stabilization. Silencing neurons with TTX stunts dendritic growth; chronic overactivation can also be harmful.
- Temperature and metabolic support: in culture, maintaining 37°C, supplying glucose and appropriate B27 supplement, and controlling CO2/pH are non-negotiable for sustained axon growth.
- Cyclic AMP levels: elevating cAMP (e.g., with forskolin or dibutyryl-cAMP) can switch a neuron's response to some guidance cues from repulsion to attraction, and generally promotes outgrowth in inhibitory environments.
In the intact brain, the same principles apply but are harder to manipulate directly. Rehabilitative activity, enriched environments, and certain pharmacological agents (including some antidepressants that elevate BDNF) have measurable effects on dendritic remodeling and synapse formation, particularly in regions like the hippocampus. This is the biological basis of experience-dependent plasticity, and understanding the growth mechanisms above tells you exactly why those interventions work.
Where to look if growth seems stuck
If you're trying to figure out which stage of neuronal growth is limiting in your system, a staged diagnostic approach is the most efficient path. Check migration and polarity first with cytoskeletal markers (MAP2 for dendrites, tau for axons). Then look at axon length and growth cone morphology to gauge whether pathfinding is occurring or stalled. Synapse formation can be assessed with pre- and postsynaptic markers (synapsin, PSD-95). If you see long axons but few synapses, the bottleneck is likely at the targeting or stabilization stage. If axons are short, the growth cone dynamics or trophic support is the problem.
This mechanistic picture also connects directly to questions about how nerves regenerate after injury, how neural networks wire themselves to become more efficient over time, and why the mature nervous system is so much more resistant to structural change than the developing one. This also helps explain how nerves grow during development, not just how they repair after injury. Those are all extensions of the same core biology described here, each adding another layer to the story of how neurons grow, stabilize, and ultimately form the networks that make thought, movement, and sensation possible. These ideas also echo in computational work on the self-assembling brain: how neural networks grow smarter. Researchers also study whether robots grow and develop in ways analogous to neural development, using adaptive materials and learning systems.
| Growth Stage | Key Players | Main Constraint |
|---|---|---|
| Polarity establishment | PI3K, PTEN, Akt, Par complex | Molecular competition; only one process wins |
| Migration | Reelin, radial glia, integrins | Stop signals and substrate availability |
| Axon extension | Growth cone actin/tubulin, guidance receptors | Energy, repulsive cues, inhibitory ECM |
| Dendrite branching | Rac1, BDNF/TrkB, activity | Activity levels, trophic support |
| Synapse formation | Neurexins, neuroligins, PSD-95 | Correlated activity, available space |
| Pruning and stabilization | Microglia, complement, neurotrophins | Competitive trophic signaling, CSPG gating |
FAQ
Do neurons start by growing an axon first, or can dendrites lead the process?
In many developmental programs the first major axon is extended before extensive dendritic branching, but the exact timing varies by neuron type and brain region. Early polarity cues still matter, so if axon identity is not established correctly, dendrites may “overgrow” and the circuit can miswire even if growth seems robust.
What determines whether a neurite becomes the axon versus a dendrite (polarity), and what goes wrong when it fails?
Polarization depends on asymmetric signaling and active cytoskeletal states, where the future axon tip supports its own growth locally and suppresses competing processes. When polarity is disrupted, neurons can generate multiple long projections or swap arbor patterns, leading to incorrect targeting and wrong layer placement.
How can an axon “choose” a direction when it receives both attractive and repulsive guidance cues at the same time?
The growth cone integrates cues by switching receptor expression and by locally reconfiguring actin and microtubule dynamics. In practice, net direction often reflects which signals dominate the GTPase balance (for example, ones that promote Rac1 or Cdc42 versus those that trigger RhoA-linked collapse), not the presence of attraction versus repulsion alone.
Why do growth cones sometimes stall even though the guidance signals are present?
Stalling can happen when the growth cone cannot sustain the cytoskeletal ratchet, for example due to insufficient membrane supply, impaired axonal transport of tubulin or actin regulators, or a mismatch between extracellular matrix composition and adhesion receptors. It can also reflect trophic support limits, where the neuron cannot maintain energy demands at the tip.
What’s the difference between pathfinding failure and synapse stabilization failure?
Pathfinding failure usually produces short or misrouted axons with growth cones that collapse or turn incorrectly. Synapse stabilization failure gives the opposite pattern, you may see contacts but little maturation, meaning reduced clustering of postsynaptic receptors and lower reinforcement despite axons reaching the target area.
If neurons grow long axons in culture, does that automatically mean functional synapses form?
No. Long axons can extend without forming stable, activity-supported connections if the environment lacks the right target cues or if activity-dependent reinforcement is absent. Functional synapse formation typically requires correct pre and postsynaptic partner identity plus correlated firing that drives reinforcement of receptor scaffolds.
How does axonal transport affect “how neurons grow” in a measurable way?
Axonal transport determines whether the growth cone receives enough building blocks, organelles, and signaling cargo to keep extending. Impairments often show up as slowed growth, frequent retractions, or degeneration of distal segments because microtubule tracks and motor-driven delivery can no longer maintain the energetic and structural needs of long projections.
Why does myelination change axon growth or maintenance?
Myelination influences conduction speed and the metabolic and signaling environment along the axon. Neurons also regulate diameter and myelination together as part of an energy budget, so an axon that is too thin or poorly supported is more likely to fail to maintain itself even if it can initially extend.
Can pruning remove “useful” connections, or is it always adaptive?
Pruning is guided by activity, competition, and immune-like removal of weak synapses, so it is generally adaptive. However, excessive or mistimed pruning (for example due to altered activity patterns) can eliminate connections that would have become useful, which is why developmental timing and experience strongly affect outcomes.
What role does the extracellular matrix play after synapses form, and can it be reversed?
The mature extracellular matrix can restrict remodeling by forming structural barriers such as perineuronal net components. In some experimental contexts, modifying specific ECM components can restore spine motility, indicating that growth restriction is not purely permanent, but rather actively enforced by the local environment.
What should I measure first if I suspect a developmental or regenerative intervention is improving neuronal growth?
Start with morphology that maps to a stage, axon length plus growth cone dynamics for outgrowth and targeting, then synapse maturation markers (not just contact frequency) for stabilization. If synapse numbers increase without longer or better-directed axons, the bottleneck is likely at stabilization or receptor recruitment rather than guidance.
Do adult neurons respond to the same growth cues as developing neurons?
Core mechanisms like guidance and activity-dependent stabilization still operate, but adults face additional brakes such as inhibitory extracellular matrix components, reduced intrinsic growth capacity, and stronger limits on structural change. That means the same cue may produce weaker extension or more retraction without the right permissive context.
Do Robots Grow and Develop? What Changes and What Doesn’t
Clear answer to do robots grow and develop, covering production changes, software learning, physical growth limits, and


